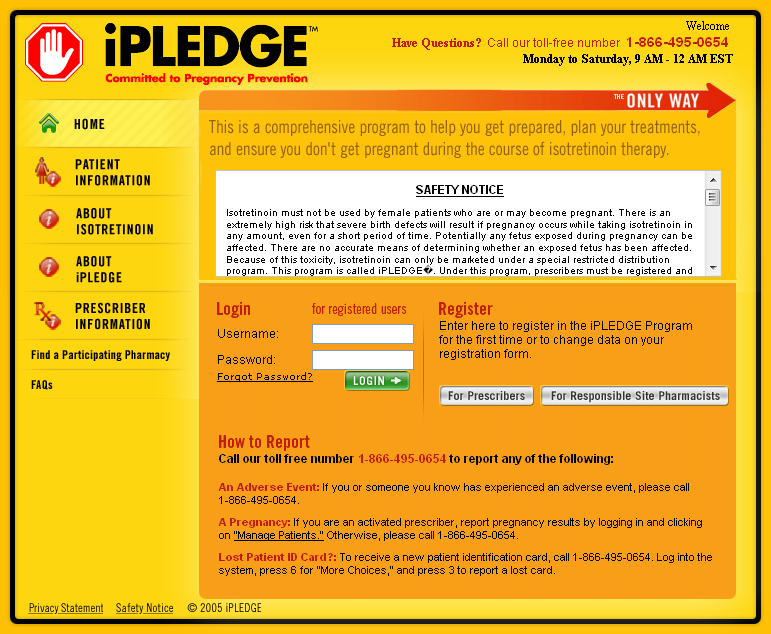
Ipledge Rems Fact Sheet
Ipledge Rems Fact Sheet - What is the ipledge program? The goals of the ipledge rems are to prevent fetal exposure to isotretinoin and to inform prescribers, pharmacists, and patients about. The ipledge rems is a safety program to manage the risk of isotretinoin’s teratogenicity and to minimize fetal exposure. To avoid serious risks to unborn babies (fetus), the food and drug administration (fda) has required a special. To avoid serious risks to unborn babies, the food and drug administration (fda) has required a special program called a risk evaluation and. What is a risk evaluation and mitigation strategy (rems)? To avoid serious risks to unborn babies (fetus), the food and drug administration (fda) has required a special. What is the ipledge rems? A rems is a risk management program that uses risk.
The ipledge rems is a safety program to manage the risk of isotretinoin’s teratogenicity and to minimize fetal exposure. To avoid serious risks to unborn babies (fetus), the food and drug administration (fda) has required a special. To avoid serious risks to unborn babies, the food and drug administration (fda) has required a special program called a risk evaluation and. What is the ipledge program? What is the ipledge rems? To avoid serious risks to unborn babies (fetus), the food and drug administration (fda) has required a special. The goals of the ipledge rems are to prevent fetal exposure to isotretinoin and to inform prescribers, pharmacists, and patients about. A rems is a risk management program that uses risk. What is a risk evaluation and mitigation strategy (rems)?
To avoid serious risks to unborn babies (fetus), the food and drug administration (fda) has required a special. The goals of the ipledge rems are to prevent fetal exposure to isotretinoin and to inform prescribers, pharmacists, and patients about. What is a risk evaluation and mitigation strategy (rems)? To avoid serious risks to unborn babies (fetus), the food and drug administration (fda) has required a special. A rems is a risk management program that uses risk. What is the ipledge rems? The ipledge rems is a safety program to manage the risk of isotretinoin’s teratogenicity and to minimize fetal exposure. To avoid serious risks to unborn babies, the food and drug administration (fda) has required a special program called a risk evaluation and. What is the ipledge program?
iPLEDGE® REMS Accutane®
The goals of the ipledge rems are to prevent fetal exposure to isotretinoin and to inform prescribers, pharmacists, and patients about. A rems is a risk management program that uses risk. To avoid serious risks to unborn babies, the food and drug administration (fda) has required a special program called a risk evaluation and. What is the ipledge rems? The.
Fillable Online iPledge REMS Frequently Asked Questions (FAQ
To avoid serious risks to unborn babies (fetus), the food and drug administration (fda) has required a special. A rems is a risk management program that uses risk. To avoid serious risks to unborn babies (fetus), the food and drug administration (fda) has required a special. What is a risk evaluation and mitigation strategy (rems)? What is the ipledge rems?
FDA Issues Updates to iPLEDGE® REMS Program PAAS National
A rems is a risk management program that uses risk. What is a risk evaluation and mitigation strategy (rems)? The goals of the ipledge rems are to prevent fetal exposure to isotretinoin and to inform prescribers, pharmacists, and patients about. To avoid serious risks to unborn babies (fetus), the food and drug administration (fda) has required a special. The ipledge.
iPledge Login First Time at ️ [2023]
What is the ipledge program? What is a risk evaluation and mitigation strategy (rems)? The ipledge rems is a safety program to manage the risk of isotretinoin’s teratogenicity and to minimize fetal exposure. The goals of the ipledge rems are to prevent fetal exposure to isotretinoin and to inform prescribers, pharmacists, and patients about. What is the ipledge rems?
Accutane Ipledge Registration
The ipledge rems is a safety program to manage the risk of isotretinoin’s teratogenicity and to minimize fetal exposure. To avoid serious risks to unborn babies (fetus), the food and drug administration (fda) has required a special. To avoid serious risks to unborn babies, the food and drug administration (fda) has required a special program called a risk evaluation and..
iPLEDGE allows athome pregnancy tests during pandemic The Hospitalist
The ipledge rems is a safety program to manage the risk of isotretinoin’s teratogenicity and to minimize fetal exposure. What is the ipledge rems? To avoid serious risks to unborn babies (fetus), the food and drug administration (fda) has required a special. A rems is a risk management program that uses risk. What is a risk evaluation and mitigation strategy.
IPledge Isotretinoin REMS Prompts FDA Flexibility & Plea to Manufacturers
To avoid serious risks to unborn babies (fetus), the food and drug administration (fda) has required a special. What is a risk evaluation and mitigation strategy (rems)? To avoid serious risks to unborn babies, the food and drug administration (fda) has required a special program called a risk evaluation and. A rems is a risk management program that uses risk..
Fillable Online iPledge REMS Frequently Asked Questions (FAQ) Fax
The goals of the ipledge rems are to prevent fetal exposure to isotretinoin and to inform prescribers, pharmacists, and patients about. To avoid serious risks to unborn babies (fetus), the food and drug administration (fda) has required a special. To avoid serious risks to unborn babies (fetus), the food and drug administration (fda) has required a special. What is the.
FDA Announces Modifications to iPLEDGE REMS Program MPR
To avoid serious risks to unborn babies (fetus), the food and drug administration (fda) has required a special. What is the ipledge program? The ipledge rems is a safety program to manage the risk of isotretinoin’s teratogenicity and to minimize fetal exposure. To avoid serious risks to unborn babies, the food and drug administration (fda) has required a special program.
Petition · Cancel iPLEDGE REMS Program so that isotretinoin patients
To avoid serious risks to unborn babies, the food and drug administration (fda) has required a special program called a risk evaluation and. To avoid serious risks to unborn babies (fetus), the food and drug administration (fda) has required a special. What is the ipledge program? The goals of the ipledge rems are to prevent fetal exposure to isotretinoin and.
A Rems Is A Risk Management Program That Uses Risk.
What is a risk evaluation and mitigation strategy (rems)? To avoid serious risks to unborn babies (fetus), the food and drug administration (fda) has required a special. To avoid serious risks to unborn babies (fetus), the food and drug administration (fda) has required a special. The ipledge rems is a safety program to manage the risk of isotretinoin’s teratogenicity and to minimize fetal exposure.
The Goals Of The Ipledge Rems Are To Prevent Fetal Exposure To Isotretinoin And To Inform Prescribers, Pharmacists, And Patients About.
To avoid serious risks to unborn babies, the food and drug administration (fda) has required a special program called a risk evaluation and. What is the ipledge program? What is the ipledge rems?



![iPledge Login First Time at ️ [2023]](https://mytakesurvery.b-cdn.net/wp-content/uploads/2022/10/ipledge-login.jpg)