Is Forming Bonds Exothermic
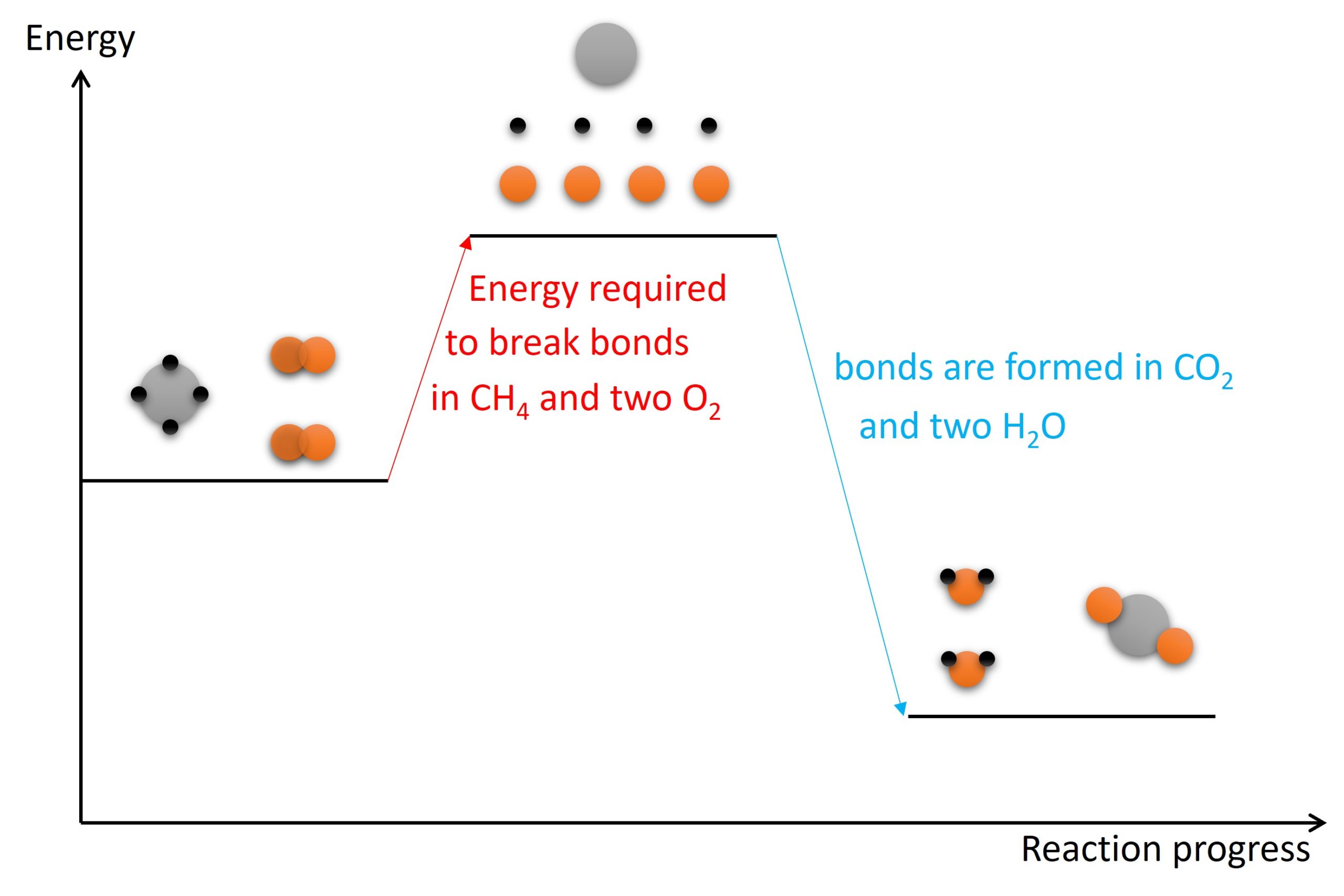
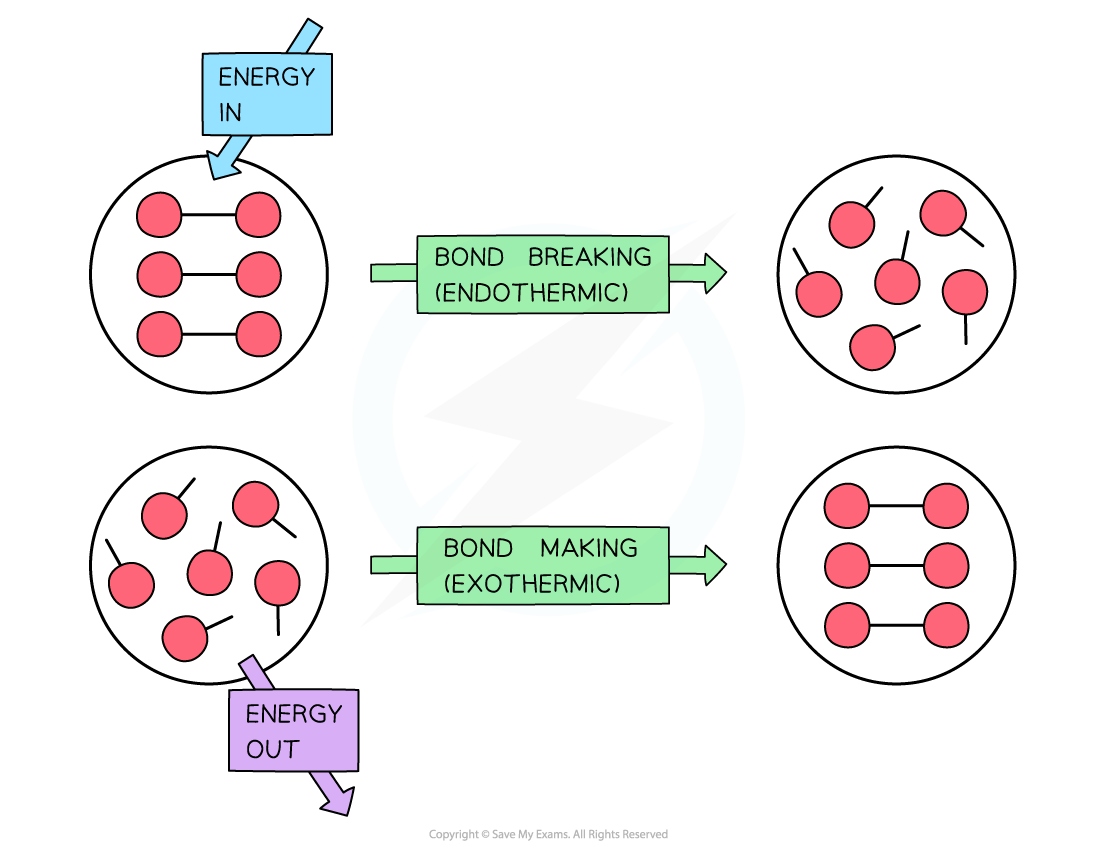
Is Forming Bonds Exothermic - Whether a reaction is endothermic or exothermic depends on the difference between the energy needed to break existing bonds and. Now that we have a basic understanding of exothermic reactions and bond enthalpy, let’s get back to the question at hand:. In summary, there are two factors which determine whether a gaseous reaction will be exothermic or not:
Now that we have a basic understanding of exothermic reactions and bond enthalpy, let’s get back to the question at hand:. Whether a reaction is endothermic or exothermic depends on the difference between the energy needed to break existing bonds and. In summary, there are two factors which determine whether a gaseous reaction will be exothermic or not:
In summary, there are two factors which determine whether a gaseous reaction will be exothermic or not: Now that we have a basic understanding of exothermic reactions and bond enthalpy, let’s get back to the question at hand:. Whether a reaction is endothermic or exothermic depends on the difference between the energy needed to break existing bonds and.
Exothermic and Endothermic Reactions and the Math Associated with Them
Now that we have a basic understanding of exothermic reactions and bond enthalpy, let’s get back to the question at hand:. Whether a reaction is endothermic or exothermic depends on the difference between the energy needed to break existing bonds and. In summary, there are two factors which determine whether a gaseous reaction will be exothermic or not:
SOLVED Which of these is the TRUE statement? A) Combustion reactions
Whether a reaction is endothermic or exothermic depends on the difference between the energy needed to break existing bonds and. Now that we have a basic understanding of exothermic reactions and bond enthalpy, let’s get back to the question at hand:. In summary, there are two factors which determine whether a gaseous reaction will be exothermic or not:
5.1 Exothermic and endothermic reactions IGCSE and A Level Chemistry
In summary, there are two factors which determine whether a gaseous reaction will be exothermic or not: Now that we have a basic understanding of exothermic reactions and bond enthalpy, let’s get back to the question at hand:. Whether a reaction is endothermic or exothermic depends on the difference between the energy needed to break existing bonds and.
SOLVED " In liquid water, water molecules are continually forming and
Whether a reaction is endothermic or exothermic depends on the difference between the energy needed to break existing bonds and. Now that we have a basic understanding of exothermic reactions and bond enthalpy, let’s get back to the question at hand:. In summary, there are two factors which determine whether a gaseous reaction will be exothermic or not:
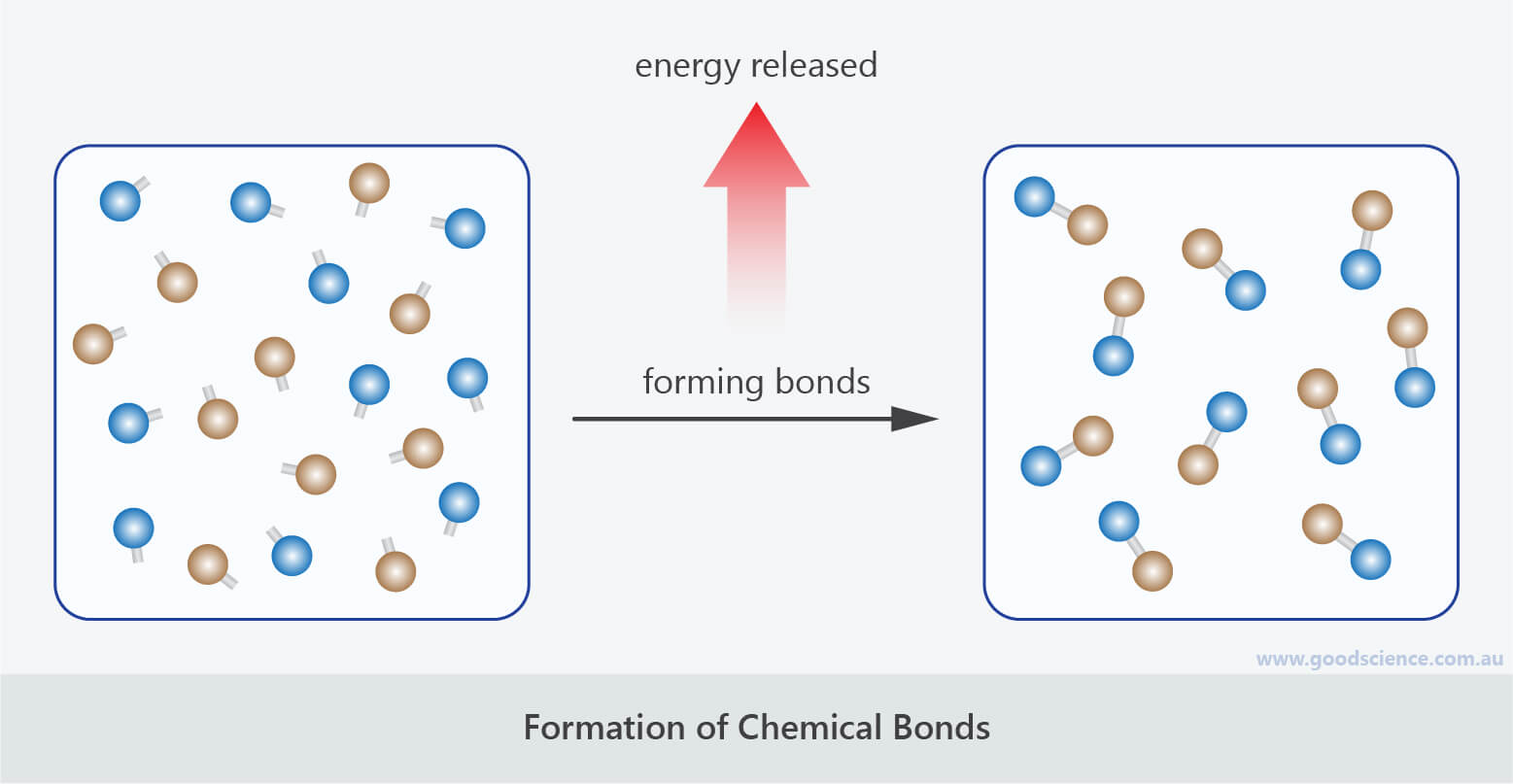
Investigating Chemical Reactions Good Science
Now that we have a basic understanding of exothermic reactions and bond enthalpy, let’s get back to the question at hand:. In summary, there are two factors which determine whether a gaseous reaction will be exothermic or not: Whether a reaction is endothermic or exothermic depends on the difference between the energy needed to break existing bonds and.
Bond energy
In summary, there are two factors which determine whether a gaseous reaction will be exothermic or not: Whether a reaction is endothermic or exothermic depends on the difference between the energy needed to break existing bonds and. Now that we have a basic understanding of exothermic reactions and bond enthalpy, let’s get back to the question at hand:.
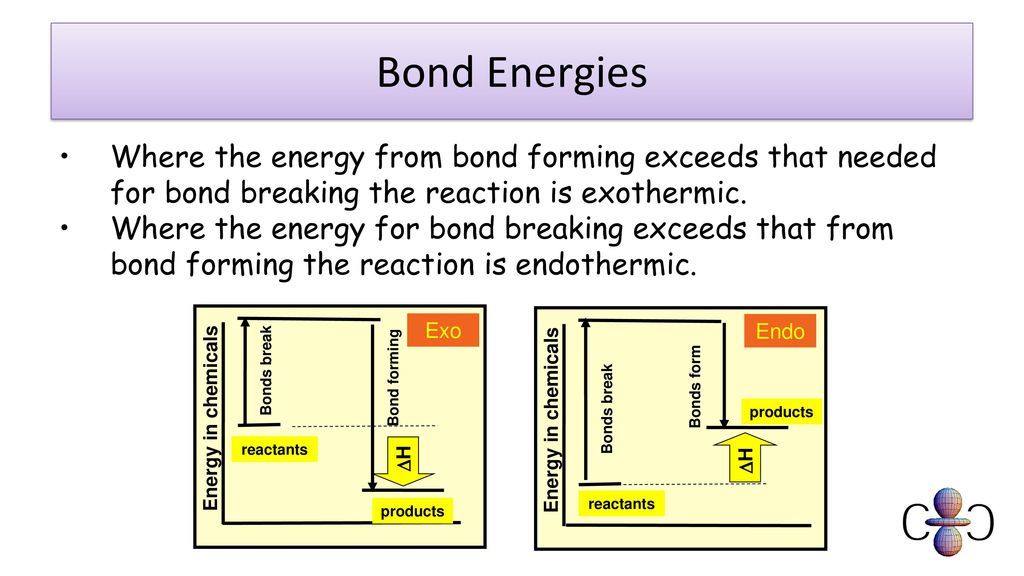
Energy changes in chemistry bond enthalpies ppt download
Now that we have a basic understanding of exothermic reactions and bond enthalpy, let’s get back to the question at hand:. Whether a reaction is endothermic or exothermic depends on the difference between the energy needed to break existing bonds and. In summary, there are two factors which determine whether a gaseous reaction will be exothermic or not:
Energetics IB Topics 5 15 PART 2 Calculating
Whether a reaction is endothermic or exothermic depends on the difference between the energy needed to break existing bonds and. In summary, there are two factors which determine whether a gaseous reaction will be exothermic or not: Now that we have a basic understanding of exothermic reactions and bond enthalpy, let’s get back to the question at hand:.
Edexcel A Level Chemistry复习笔记1.8.5 Bond Enthalpy翰林国际教育
In summary, there are two factors which determine whether a gaseous reaction will be exothermic or not: Whether a reaction is endothermic or exothermic depends on the difference between the energy needed to break existing bonds and. Now that we have a basic understanding of exothermic reactions and bond enthalpy, let’s get back to the question at hand:.
PPT Energetics PowerPoint Presentation, free download ID9217507
Now that we have a basic understanding of exothermic reactions and bond enthalpy, let’s get back to the question at hand:. In summary, there are two factors which determine whether a gaseous reaction will be exothermic or not: Whether a reaction is endothermic or exothermic depends on the difference between the energy needed to break existing bonds and.
Whether A Reaction Is Endothermic Or Exothermic Depends On The Difference Between The Energy Needed To Break Existing Bonds And.
In summary, there are two factors which determine whether a gaseous reaction will be exothermic or not: Now that we have a basic understanding of exothermic reactions and bond enthalpy, let’s get back to the question at hand:.