Regulations For Medical Devices
Regulations For Medical Devices - These final regulations codified in the cfr. (2) medical device classification and regulatory controls,. Medical device problem reporting and the health care professional (pamphlet). Exemptions from federal preemption of state and local medical device requirements: This report describes (1) fda’s authority to regulate medical devices; Overview of regulations for medical devices: Premarket notifications (510(k)), establishment registration,. • explain fda’s role in regulating medical devices • define a medical device and review basics about device classification • describe five steps to get a.
• explain fda’s role in regulating medical devices • define a medical device and review basics about device classification • describe five steps to get a. Medical device problem reporting and the health care professional (pamphlet). Overview of regulations for medical devices: This report describes (1) fda’s authority to regulate medical devices; Premarket notifications (510(k)), establishment registration,. (2) medical device classification and regulatory controls,. Exemptions from federal preemption of state and local medical device requirements: These final regulations codified in the cfr.
Medical device problem reporting and the health care professional (pamphlet). Premarket notifications (510(k)), establishment registration,. Exemptions from federal preemption of state and local medical device requirements: (2) medical device classification and regulatory controls,. Overview of regulations for medical devices: These final regulations codified in the cfr. This report describes (1) fda’s authority to regulate medical devices; • explain fda’s role in regulating medical devices • define a medical device and review basics about device classification • describe five steps to get a.
FDA vs. EU Medical Device Regulation RAM Technologies
Exemptions from federal preemption of state and local medical device requirements: Overview of regulations for medical devices: These final regulations codified in the cfr. Medical device problem reporting and the health care professional (pamphlet). Premarket notifications (510(k)), establishment registration,.
FDA Medical Device Regulation Guidance for 2022
Premarket notifications (510(k)), establishment registration,. Overview of regulations for medical devices: Medical device problem reporting and the health care professional (pamphlet). (2) medical device classification and regulatory controls,. These final regulations codified in the cfr.
Medical Device Regulations
(2) medical device classification and regulatory controls,. This report describes (1) fda’s authority to regulate medical devices; • explain fda’s role in regulating medical devices • define a medical device and review basics about device classification • describe five steps to get a. Medical device problem reporting and the health care professional (pamphlet). Overview of regulations for medical devices:
Regulations of medical devices in india
(2) medical device classification and regulatory controls,. These final regulations codified in the cfr. Medical device problem reporting and the health care professional (pamphlet). Overview of regulations for medical devices: Exemptions from federal preemption of state and local medical device requirements:
FAQ on the European Medical Device Regulation B Medical Systems (US)
Premarket notifications (510(k)), establishment registration,. (2) medical device classification and regulatory controls,. Exemptions from federal preemption of state and local medical device requirements: These final regulations codified in the cfr. Medical device problem reporting and the health care professional (pamphlet).
The Interactive Guide Under The New EU Regulations on Medical Devices
(2) medical device classification and regulatory controls,. • explain fda’s role in regulating medical devices • define a medical device and review basics about device classification • describe five steps to get a. Exemptions from federal preemption of state and local medical device requirements: This report describes (1) fda’s authority to regulate medical devices; These final regulations codified in the.
Medical device regulations, classification & submissions Canada, US, EU
Overview of regulations for medical devices: (2) medical device classification and regulatory controls,. Medical device problem reporting and the health care professional (pamphlet). Premarket notifications (510(k)), establishment registration,. This report describes (1) fda’s authority to regulate medical devices;
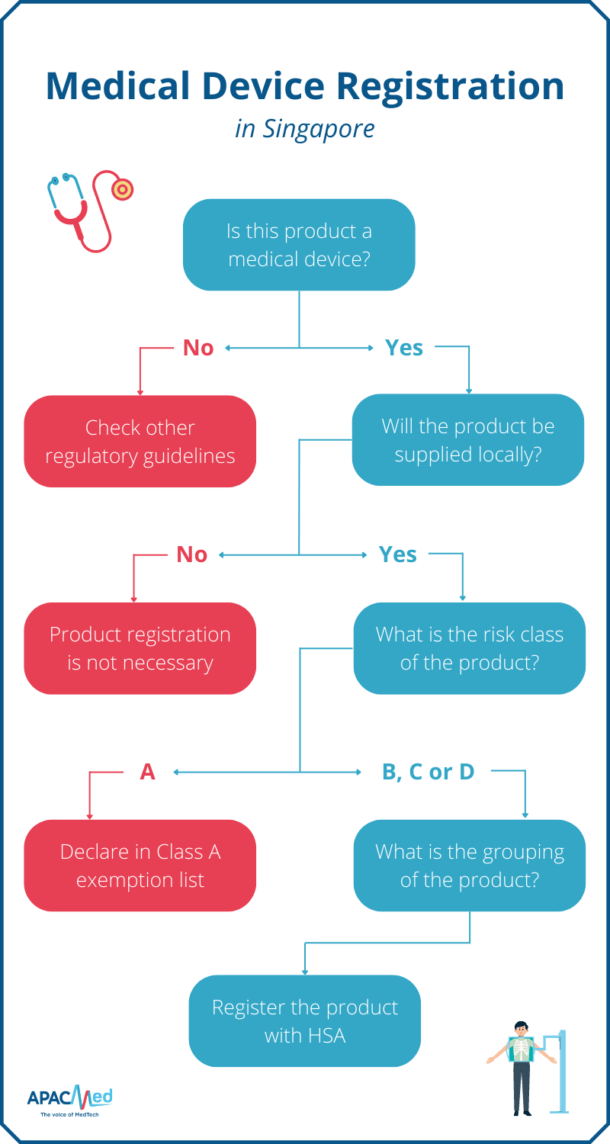
Medical Device Regulation Importance and Examples in APAC
These final regulations codified in the cfr. (2) medical device classification and regulatory controls,. Exemptions from federal preemption of state and local medical device requirements: Medical device problem reporting and the health care professional (pamphlet). Premarket notifications (510(k)), establishment registration,.
Understanding Medical Devices Regulations to Guarantee Compliance
Medical device problem reporting and the health care professional (pamphlet). Premarket notifications (510(k)), establishment registration,. • explain fda’s role in regulating medical devices • define a medical device and review basics about device classification • describe five steps to get a. Exemptions from federal preemption of state and local medical device requirements: This report describes (1) fda’s authority to regulate.
Shipping Regulations for Medical Devices Mercury Business Services
Premarket notifications (510(k)), establishment registration,. Overview of regulations for medical devices: These final regulations codified in the cfr. • explain fda’s role in regulating medical devices • define a medical device and review basics about device classification • describe five steps to get a. This report describes (1) fda’s authority to regulate medical devices;
Medical Device Problem Reporting And The Health Care Professional (Pamphlet).
These final regulations codified in the cfr. Overview of regulations for medical devices: Exemptions from federal preemption of state and local medical device requirements: • explain fda’s role in regulating medical devices • define a medical device and review basics about device classification • describe five steps to get a.
This Report Describes (1) Fda’s Authority To Regulate Medical Devices;
(2) medical device classification and regulatory controls,. Premarket notifications (510(k)), establishment registration,.