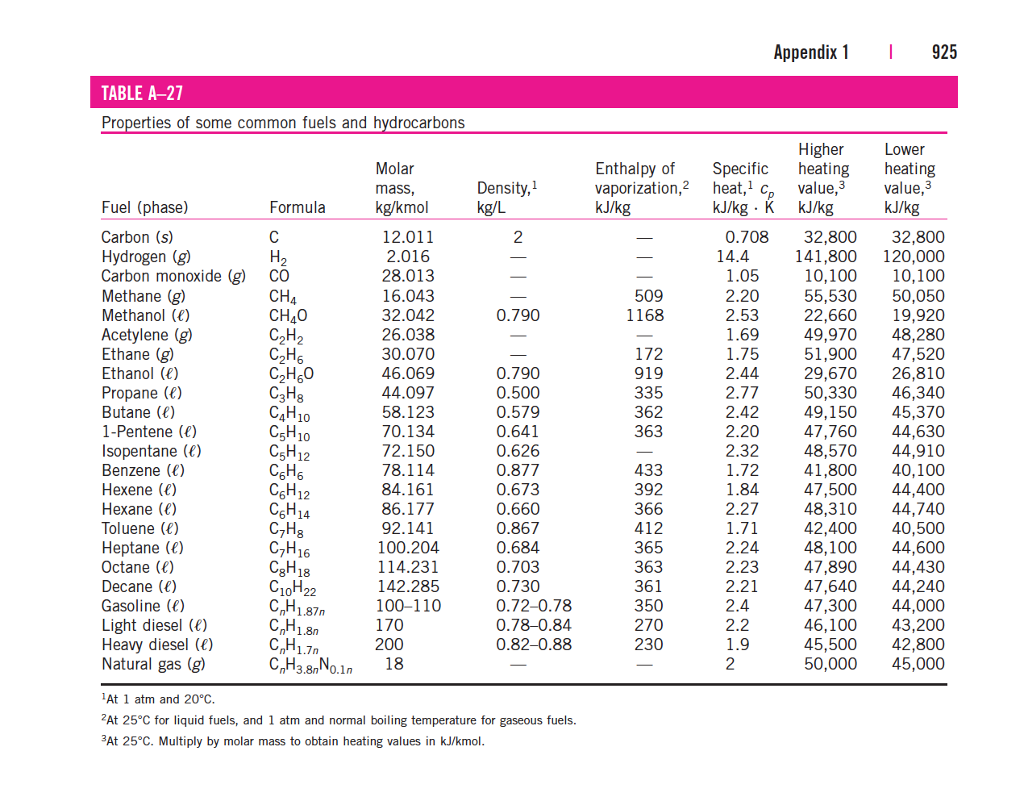
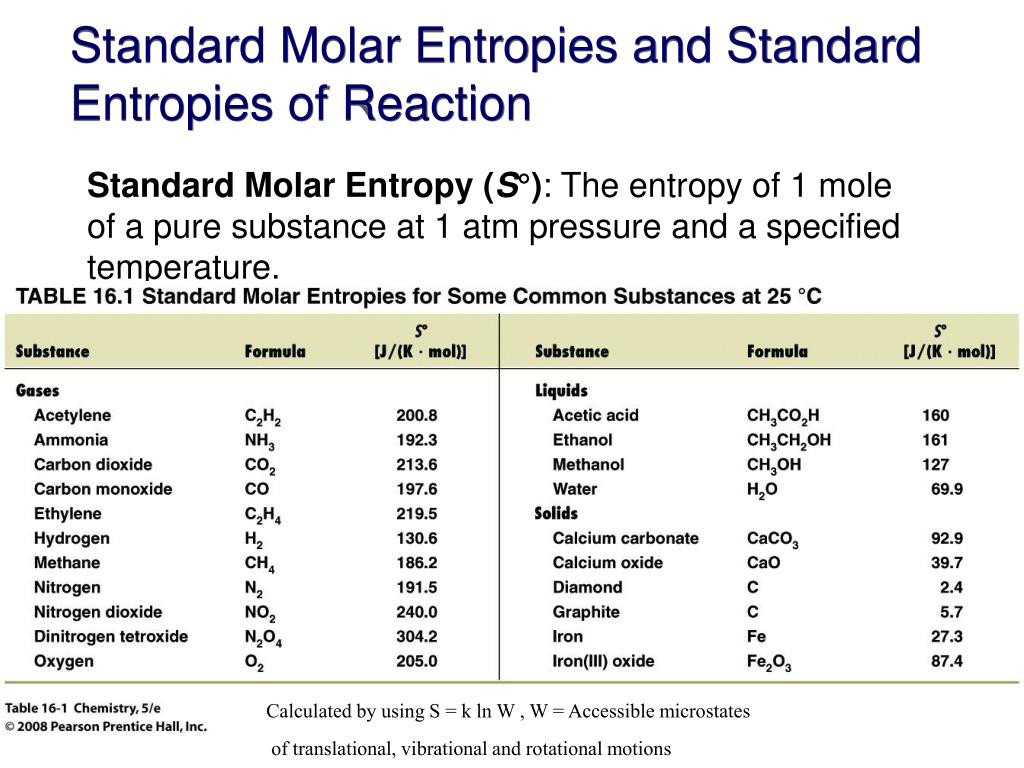
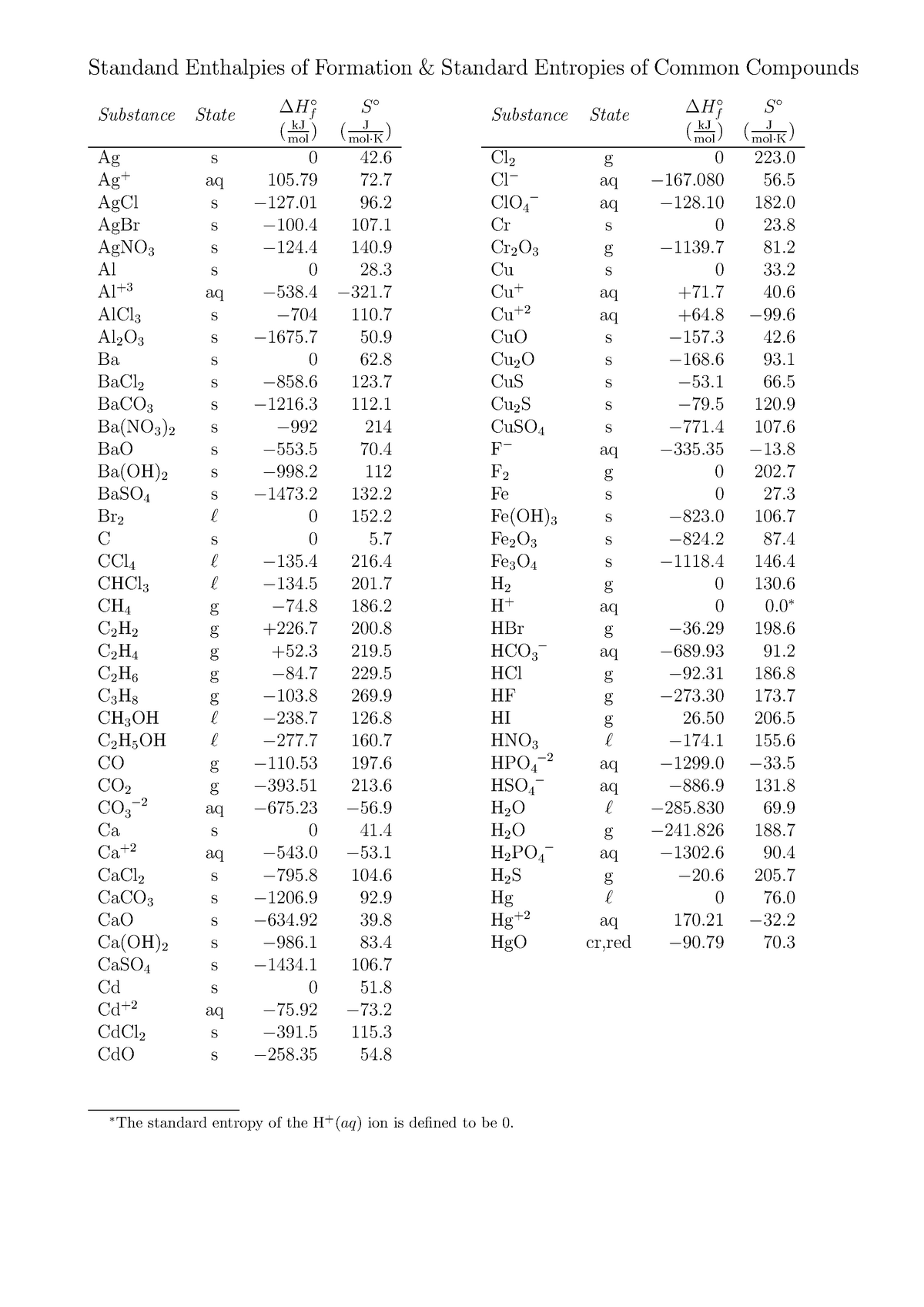
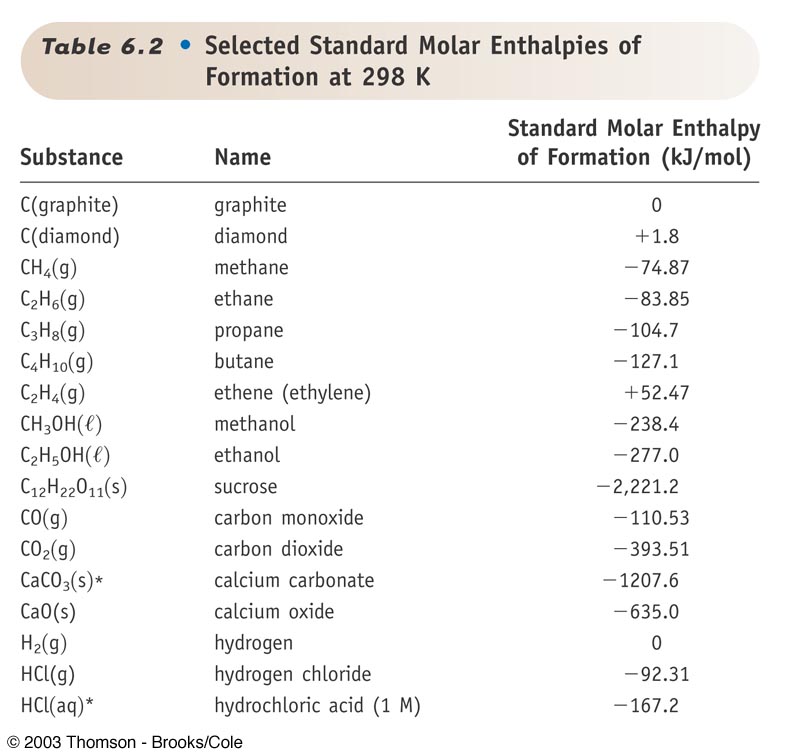
Standard Entropy Of Formation Table
Standard Entropy Of Formation Table - Compound state δh f ° (kj/mol) δg f ° (kj/mol) s° (j/mol k) class; The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds from elements in their standard states, and the. 100 rows standard heats and free energies of formation and absolute entropies of elements and inorganic compounds ∗the standard entropy of the h+(aq) ion is defined to be 0.
100 rows standard heats and free energies of formation and absolute entropies of elements and inorganic compounds ∗the standard entropy of the h+(aq) ion is defined to be 0. This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds from elements in their standard states, and the. Compound state δh f ° (kj/mol) δg f ° (kj/mol) s° (j/mol k) class; The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat.
100 rows standard heats and free energies of formation and absolute entropies of elements and inorganic compounds ∗the standard entropy of the h+(aq) ion is defined to be 0. Compound state δh f ° (kj/mol) δg f ° (kj/mol) s° (j/mol k) class; The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds from elements in their standard states, and the.
TABLE A286 Enthalpy of formation, Gibbs function of
∗the standard entropy of the h+(aq) ion is defined to be 0. Compound state δh f ° (kj/mol) δg f ° (kj/mol) s° (j/mol k) class; The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. 100 rows standard heats and free energies of formation and absolute entropies of.
1. For the reaction Hg(1) + O2 → HgO(s) Use absolute entropies from
The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. 100 rows standard heats and free energies of formation and absolute entropies of elements and inorganic compounds This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds from elements in their standard states, and.
Table II from Largescale calculations of gas phase thermochemistry
Compound state δh f ° (kj/mol) δg f ° (kj/mol) s° (j/mol k) class; 100 rows standard heats and free energies of formation and absolute entropies of elements and inorganic compounds ∗the standard entropy of the h+(aq) ion is defined to be 0. The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard.
Astounding Collections Of Heat Of Formation Table Photos Darkata
The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. 100 rows standard heats and free energies of formation and absolute entropies of elements and inorganic compounds This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds from elements in their standard states, and.
Standard entropy wsmzaer
Compound state δh f ° (kj/mol) δg f ° (kj/mol) s° (j/mol k) class; This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds from elements in their standard states, and the. The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. ∗the standard.
Table I from Largescale calculations of gas phase thermochemistry
The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds from elements in their standard states, and the. Compound state δh f ° (kj/mol) δg f ° (kj/mol) s° (j/mol k) class; ∗the standard.
Standard entropy table Lasialabama
The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds from elements in their standard states, and the. ∗the standard entropy of the h+(aq) ion is defined to be 0. Compound state δh f.
PPT Entropy PowerPoint Presentation, free download ID2017581
This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds from elements in their standard states, and the. Compound state δh f ° (kj/mol) δg f ° (kj/mol) s° (j/mol k) class; 100 rows standard heats and free energies of formation and absolute entropies of elements and inorganic compounds ∗the standard entropy of the h+(aq).
Heat Of Formation Chart
The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. Compound state δh f ° (kj/mol) δg f ° (kj/mol) s° (j/mol k) class; This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds from elements in their standard states, and the. ∗the standard.
Change in Enthalpy Chemistry lessons, Chemistry, Ap chemistry
The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. Compound state δh f ° (kj/mol) δg f ° (kj/mol) s° (j/mol k) class; 100 rows standard heats and free energies of formation and absolute entropies of elements and inorganic compounds This table lists the standard enthalpies (δh°), the.
This Table Lists The Standard Enthalpies (Δh°), The Free Energies (Δg°) Of Formation Of Compounds From Elements In Their Standard States, And The.
∗the standard entropy of the h+(aq) ion is defined to be 0. 100 rows standard heats and free energies of formation and absolute entropies of elements and inorganic compounds The table below shows the standard enthalpy of formation, the standard gibbs free energy of formation, standard entropy and molar heat. Compound state δh f ° (kj/mol) δg f ° (kj/mol) s° (j/mol k) class;