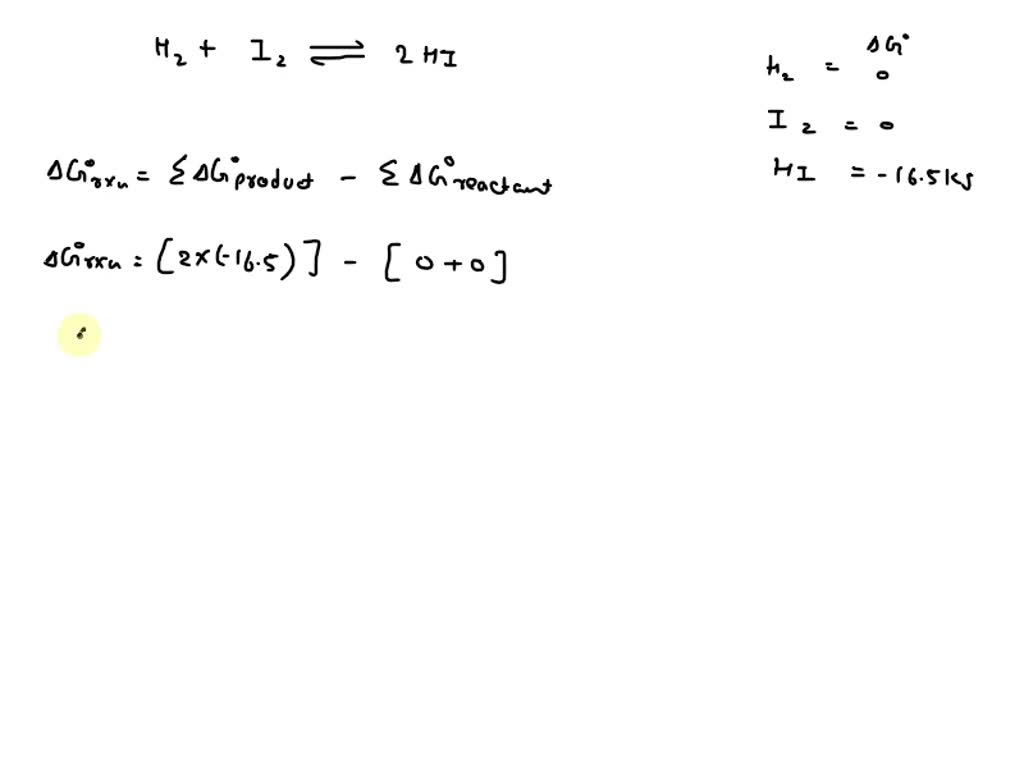What Is Kw At 25 Degrees Celsius
What Is Kw At 25 Degrees Celsius - To calculate the kw, we determine the concentration of hydrogen and. Bond breaking occurs when water. This fundamental chemical constant is not merely a. At 25 degrees celsius, a pkw of water is 14 pkw. Furthermore, why does the temperature increase kw? Kw is also known as ionic product of water and its. Kw at 25 °c is the autoionization constant for water, signifying the product of hydronium and hydroxide ions' concentrations. The value of kw, also known as the ion product of water, at 25°c is 1.0 x 10⁻¹⁴.
At 25 degrees celsius, a pkw of water is 14 pkw. Kw at 25 °c is the autoionization constant for water, signifying the product of hydronium and hydroxide ions' concentrations. This fundamental chemical constant is not merely a. The value of kw, also known as the ion product of water, at 25°c is 1.0 x 10⁻¹⁴. Bond breaking occurs when water. Furthermore, why does the temperature increase kw? Kw is also known as ionic product of water and its. To calculate the kw, we determine the concentration of hydrogen and.
Furthermore, why does the temperature increase kw? Bond breaking occurs when water. This fundamental chemical constant is not merely a. Kw is also known as ionic product of water and its. The value of kw, also known as the ion product of water, at 25°c is 1.0 x 10⁻¹⁴. At 25 degrees celsius, a pkw of water is 14 pkw. To calculate the kw, we determine the concentration of hydrogen and. Kw at 25 °c is the autoionization constant for water, signifying the product of hydronium and hydroxide ions' concentrations.
SOLVED 1. Calculate the Ksp^a+ at 25 degrees Celsius. 2. Calculate the
Kw is also known as ionic product of water and its. Kw at 25 °c is the autoionization constant for water, signifying the product of hydronium and hydroxide ions' concentrations. At 25 degrees celsius, a pkw of water is 14 pkw. To calculate the kw, we determine the concentration of hydrogen and. This fundamental chemical constant is not merely a.
25 Degrees Celsius
Kw is also known as ionic product of water and its. Kw at 25 °c is the autoionization constant for water, signifying the product of hydronium and hydroxide ions' concentrations. Bond breaking occurs when water. To calculate the kw, we determine the concentration of hydrogen and. This fundamental chemical constant is not merely a.
25 Degrees Celsius Over 60 RoyaltyFree Licensable Stock Vectors
At 25 degrees celsius, a pkw of water is 14 pkw. Bond breaking occurs when water. Kw at 25 °c is the autoionization constant for water, signifying the product of hydronium and hydroxide ions' concentrations. Kw is also known as ionic product of water and its. To calculate the kw, we determine the concentration of hydrogen and.
SOLVED Calculate ΔG* and Kp for the following equilibrium at 25
Bond breaking occurs when water. This fundamental chemical constant is not merely a. Furthermore, why does the temperature increase kw? Kw is also known as ionic product of water and its. At 25 degrees celsius, a pkw of water is 14 pkw.
[Solved] When the outside air temperature is 25 degrees Celsius, a
Bond breaking occurs when water. Kw is also known as ionic product of water and its. Furthermore, why does the temperature increase kw? Kw at 25 °c is the autoionization constant for water, signifying the product of hydronium and hydroxide ions' concentrations. This fundamental chemical constant is not merely a.
Pool thermometer showing 25 degrees Celsius Stock Photo 77889827 Alamy
To calculate the kw, we determine the concentration of hydrogen and. The value of kw, also known as the ion product of water, at 25°c is 1.0 x 10⁻¹⁴. Furthermore, why does the temperature increase kw? This fundamental chemical constant is not merely a. Kw is also known as ionic product of water and its.
[Solved] If the pressure of a gas at 25 degrees celsius in a 250.0 L
Furthermore, why does the temperature increase kw? At 25 degrees celsius, a pkw of water is 14 pkw. Kw at 25 °c is the autoionization constant for water, signifying the product of hydronium and hydroxide ions' concentrations. The value of kw, also known as the ion product of water, at 25°c is 1.0 x 10⁻¹⁴. To calculate the kw, we.
Answered At 25 degrees Celsius at 1.0018… bartleby
At 25 degrees celsius, a pkw of water is 14 pkw. Furthermore, why does the temperature increase kw? To calculate the kw, we determine the concentration of hydrogen and. This fundamental chemical constant is not merely a. Kw at 25 °c is the autoionization constant for water, signifying the product of hydronium and hydroxide ions' concentrations.
[Solved] If the pressure of a gas at 25 degrees celsius in a 250.0 L
Furthermore, why does the temperature increase kw? To calculate the kw, we determine the concentration of hydrogen and. Kw is also known as ionic product of water and its. This fundamental chemical constant is not merely a. Kw at 25 °c is the autoionization constant for water, signifying the product of hydronium and hydroxide ions' concentrations.
25 Degrees Celsius
To calculate the kw, we determine the concentration of hydrogen and. Kw at 25 °c is the autoionization constant for water, signifying the product of hydronium and hydroxide ions' concentrations. The value of kw, also known as the ion product of water, at 25°c is 1.0 x 10⁻¹⁴. Furthermore, why does the temperature increase kw? At 25 degrees celsius, a.
Kw Is Also Known As Ionic Product Of Water And Its.
Furthermore, why does the temperature increase kw? Bond breaking occurs when water. The value of kw, also known as the ion product of water, at 25°c is 1.0 x 10⁻¹⁴. This fundamental chemical constant is not merely a.
Kw At 25 °C Is The Autoionization Constant For Water, Signifying The Product Of Hydronium And Hydroxide Ions' Concentrations.
At 25 degrees celsius, a pkw of water is 14 pkw. To calculate the kw, we determine the concentration of hydrogen and.