What Is The Electron Configuration Of Cu2
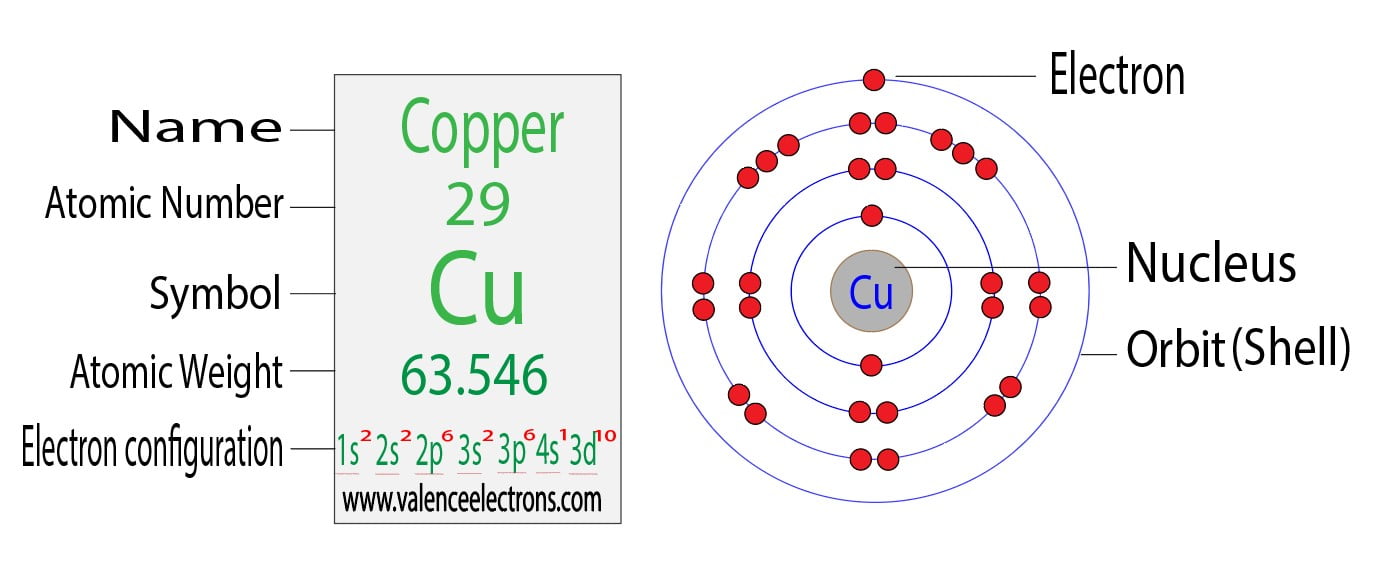
What Is The Electron Configuration Of Cu2 - It loses two electrons from the 4s and. Full electron configuration (same as co) #1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^7# noble gas electron configuration #[ar]4s^2. How to write the electron configuration for copper (cu, cu+, and cu2+) in order to write the copper electron configuration we first need to know the. The correct electron configuration for the cu2+ ion is [ar] 3d9. To determine the electron configuration of cu2+, which represents a copper ion with a +2 charge, we need to determine its electron configuration. What is the correct electron configuration for the cu2+ ion?
How to write the electron configuration for copper (cu, cu+, and cu2+) in order to write the copper electron configuration we first need to know the. The correct electron configuration for the cu2+ ion is [ar] 3d9. It loses two electrons from the 4s and. To determine the electron configuration of cu2+, which represents a copper ion with a +2 charge, we need to determine its electron configuration. Full electron configuration (same as co) #1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^7# noble gas electron configuration #[ar]4s^2. What is the correct electron configuration for the cu2+ ion?
What is the correct electron configuration for the cu2+ ion? It loses two electrons from the 4s and. Full electron configuration (same as co) #1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^7# noble gas electron configuration #[ar]4s^2. The correct electron configuration for the cu2+ ion is [ar] 3d9. How to write the electron configuration for copper (cu, cu+, and cu2+) in order to write the copper electron configuration we first need to know the. To determine the electron configuration of cu2+, which represents a copper ion with a +2 charge, we need to determine its electron configuration.
What is Electron Configuration for Cu+2? ResearchGate
Full electron configuration (same as co) #1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^7# noble gas electron configuration #[ar]4s^2. How to write the electron configuration for copper (cu, cu+, and cu2+) in order to write the copper electron configuration we first need to know the. What is the correct electron configuration for the cu2+ ion? The correct electron configuration for the.
Copper Electron Configuration (Cu) with Orbital Diagram
The correct electron configuration for the cu2+ ion is [ar] 3d9. To determine the electron configuration of cu2+, which represents a copper ion with a +2 charge, we need to determine its electron configuration. It loses two electrons from the 4s and. Full electron configuration (same as co) #1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^7# noble gas electron configuration #[ar]4s^2..
Electron Configuration Of Copper
It loses two electrons from the 4s and. The correct electron configuration for the cu2+ ion is [ar] 3d9. To determine the electron configuration of cu2+, which represents a copper ion with a +2 charge, we need to determine its electron configuration. What is the correct electron configuration for the cu2+ ion? How to write the electron configuration for copper.
65 CR 2+ ELECTRON CONFIGURATION * ElectronConfiguration
How to write the electron configuration for copper (cu, cu+, and cu2+) in order to write the copper electron configuration we first need to know the. The correct electron configuration for the cu2+ ion is [ar] 3d9. What is the correct electron configuration for the cu2+ ion? To determine the electron configuration of cu2+, which represents a copper ion with.
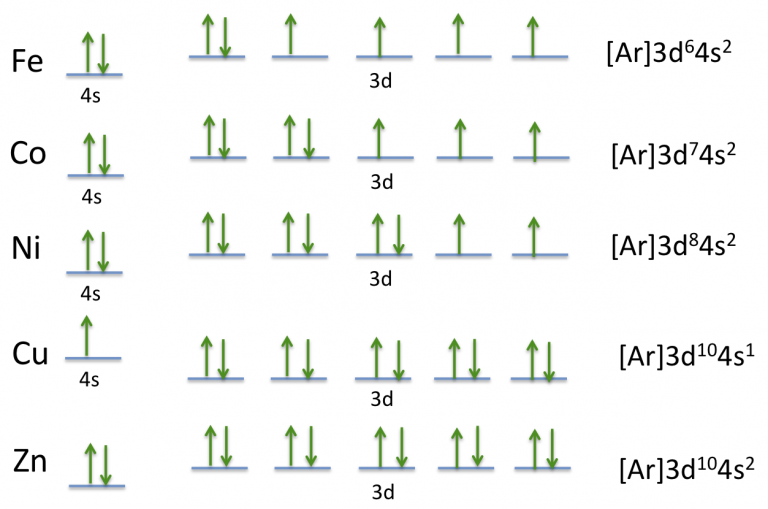
Cobalt Electron Configuration (Co) with Orbital Diagram
The correct electron configuration for the cu2+ ion is [ar] 3d9. It loses two electrons from the 4s and. How to write the electron configuration for copper (cu, cu+, and cu2+) in order to write the copper electron configuration we first need to know the. What is the correct electron configuration for the cu2+ ion? To determine the electron configuration.
Schematic Diagram Of The Electron Configuration Aluminum Orbital
How to write the electron configuration for copper (cu, cu+, and cu2+) in order to write the copper electron configuration we first need to know the. To determine the electron configuration of cu2+, which represents a copper ion with a +2 charge, we need to determine its electron configuration. It loses two electrons from the 4s and. Full electron configuration.
Gallium(Ga) electron configuration and orbital diagram
To determine the electron configuration of cu2+, which represents a copper ion with a +2 charge, we need to determine its electron configuration. The correct electron configuration for the cu2+ ion is [ar] 3d9. What is the correct electron configuration for the cu2+ ion? It loses two electrons from the 4s and. Full electron configuration (same as co) #1s^2 2s^2.
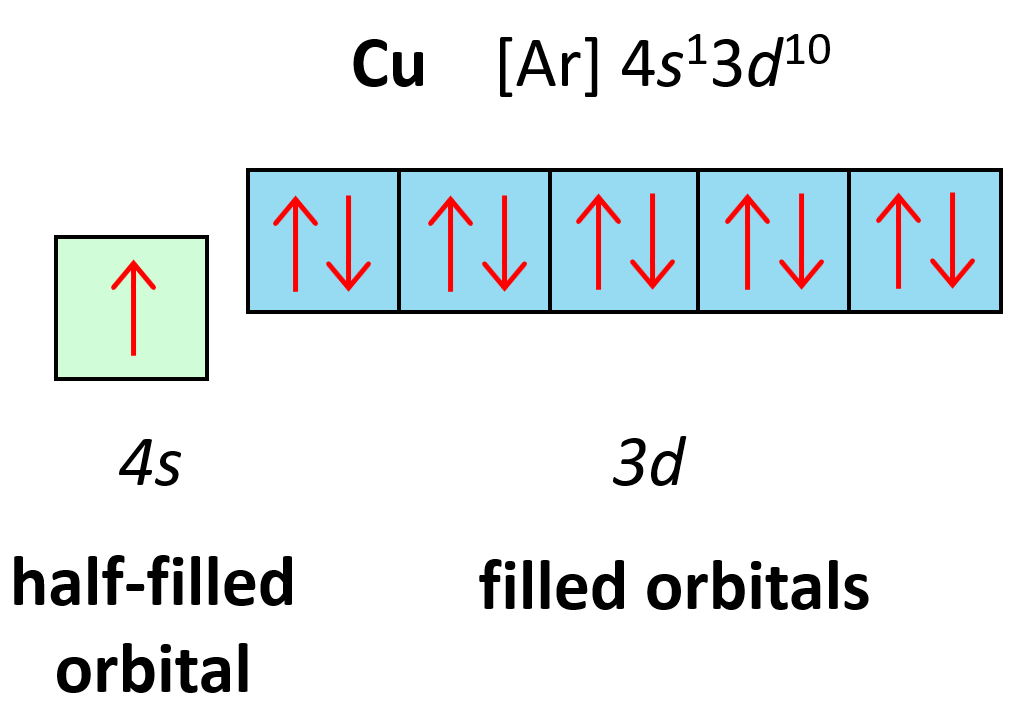
Electron Configuration for Copper (Cu, Cu+, Cu2+)
How to write the electron configuration for copper (cu, cu+, and cu2+) in order to write the copper electron configuration we first need to know the. What is the correct electron configuration for the cu2+ ion? It loses two electrons from the 4s and. The correct electron configuration for the cu2+ ion is [ar] 3d9. Full electron configuration (same as.
Electron Configuration for Copper (Cu, Cu+, Cu2+)
Full electron configuration (same as co) #1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^7# noble gas electron configuration #[ar]4s^2. It loses two electrons from the 4s and. How to write the electron configuration for copper (cu, cu+, and cu2+) in order to write the copper electron configuration we first need to know the. What is the correct electron configuration for the.
Electron Configuration for Copper (Cu, Cu+, Cu2+)
What is the correct electron configuration for the cu2+ ion? The correct electron configuration for the cu2+ ion is [ar] 3d9. How to write the electron configuration for copper (cu, cu+, and cu2+) in order to write the copper electron configuration we first need to know the. Full electron configuration (same as co) #1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^7#.
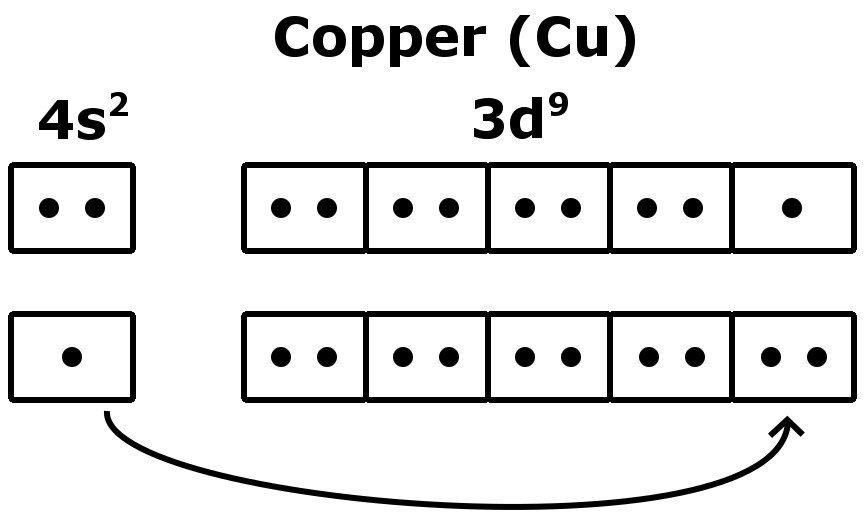
It Loses Two Electrons From The 4S And.
Full electron configuration (same as co) #1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^7# noble gas electron configuration #[ar]4s^2. How to write the electron configuration for copper (cu, cu+, and cu2+) in order to write the copper electron configuration we first need to know the. What is the correct electron configuration for the cu2+ ion? The correct electron configuration for the cu2+ ion is [ar] 3d9.