What Is The Hybridization Of Nh4
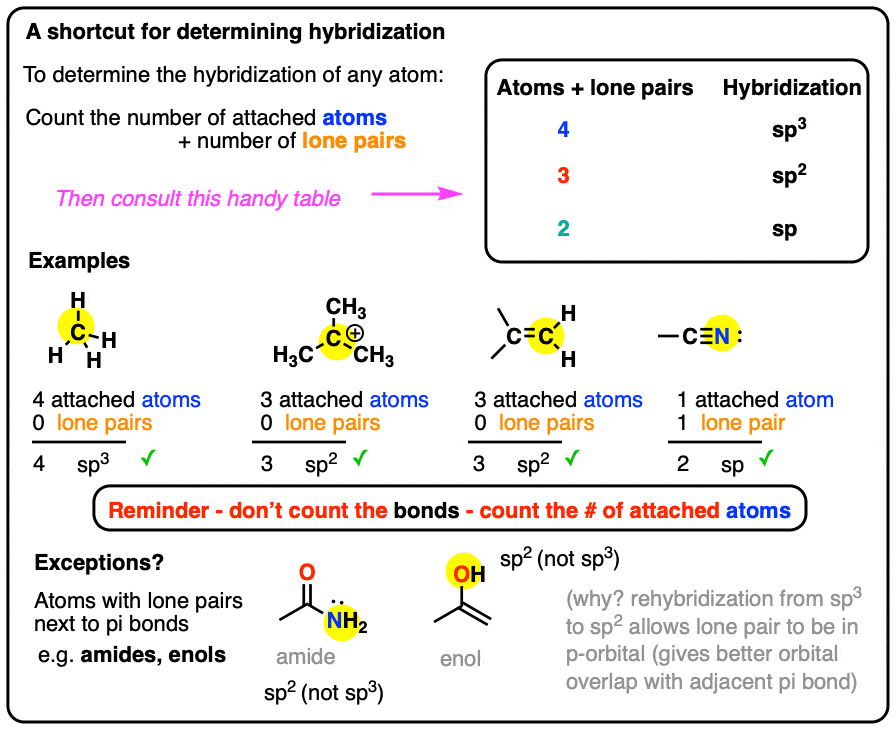
What Is The Hybridization Of Nh4 - The nitrogen atom is attached, to three h atoms through three sigma covalent bonds and to one h^+ ion through one sigma. According to vsepr theory we can find the hybridization of atoms in a molecule by the summation of the number of lone pairs of. The electronic configuration of a nitrogen (n) atom is 1s 2 2s 2. The central nitrogen atom has sp 3 hybridization in the [nh 4] + ion.
The electronic configuration of a nitrogen (n) atom is 1s 2 2s 2. The nitrogen atom is attached, to three h atoms through three sigma covalent bonds and to one h^+ ion through one sigma. The central nitrogen atom has sp 3 hybridization in the [nh 4] + ion. According to vsepr theory we can find the hybridization of atoms in a molecule by the summation of the number of lone pairs of.
According to vsepr theory we can find the hybridization of atoms in a molecule by the summation of the number of lone pairs of. The central nitrogen atom has sp 3 hybridization in the [nh 4] + ion. The electronic configuration of a nitrogen (n) atom is 1s 2 2s 2. The nitrogen atom is attached, to three h atoms through three sigma covalent bonds and to one h^+ ion through one sigma.
What are the Important Characteristics of Hybridization? Aakash BYJU
The nitrogen atom is attached, to three h atoms through three sigma covalent bonds and to one h^+ ion through one sigma. The electronic configuration of a nitrogen (n) atom is 1s 2 2s 2. The central nitrogen atom has sp 3 hybridization in the [nh 4] + ion. According to vsepr theory we can find the hybridization of atoms.
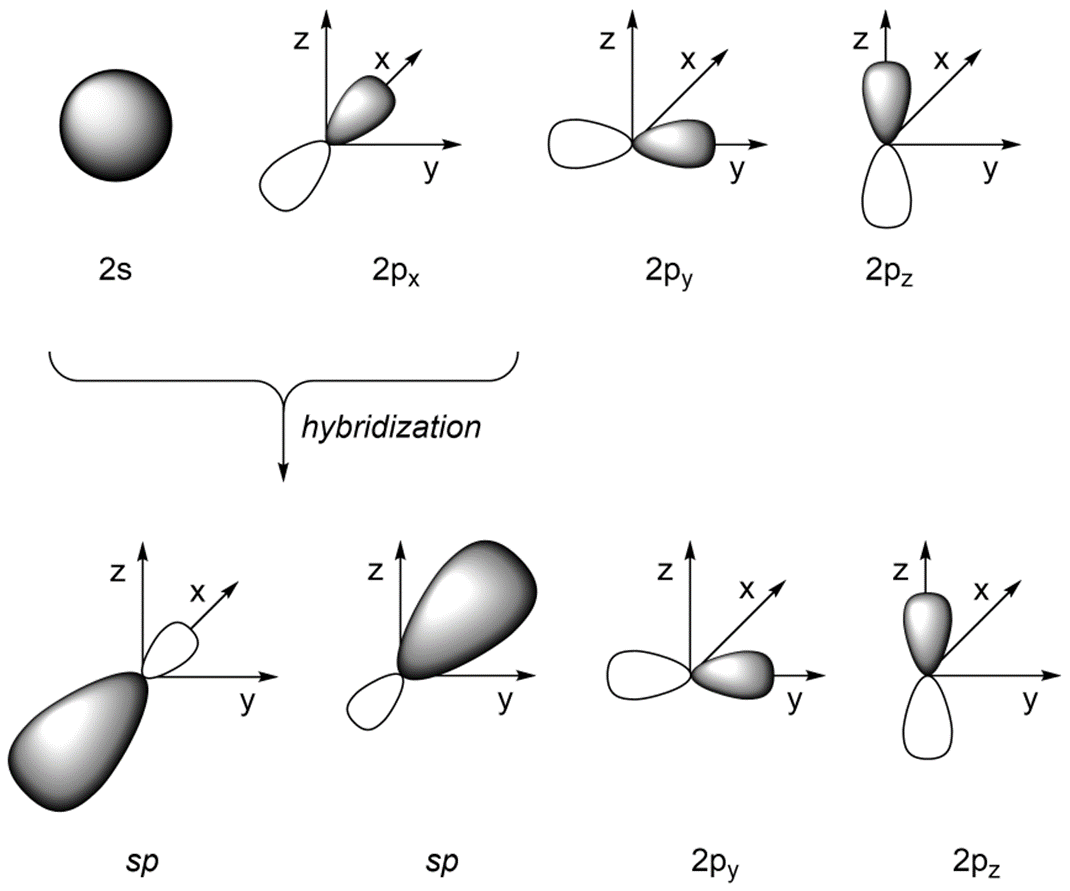
Hybridization Chart
The electronic configuration of a nitrogen (n) atom is 1s 2 2s 2. According to vsepr theory we can find the hybridization of atoms in a molecule by the summation of the number of lone pairs of. The central nitrogen atom has sp 3 hybridization in the [nh 4] + ion. The nitrogen atom is attached, to three h atoms.
Hybridization Orbitals Chart
The nitrogen atom is attached, to three h atoms through three sigma covalent bonds and to one h^+ ion through one sigma. The central nitrogen atom has sp 3 hybridization in the [nh 4] + ion. According to vsepr theory we can find the hybridization of atoms in a molecule by the summation of the number of lone pairs of..
NH4+ lewis structure, molecular geometry, bond angle, hybridization
The electronic configuration of a nitrogen (n) atom is 1s 2 2s 2. According to vsepr theory we can find the hybridization of atoms in a molecule by the summation of the number of lone pairs of. The nitrogen atom is attached, to three h atoms through three sigma covalent bonds and to one h^+ ion through one sigma. The.
Hybridization Chemistry
According to vsepr theory we can find the hybridization of atoms in a molecule by the summation of the number of lone pairs of. The electronic configuration of a nitrogen (n) atom is 1s 2 2s 2. The nitrogen atom is attached, to three h atoms through three sigma covalent bonds and to one h^+ ion through one sigma. The.
Hybridization Definition, Types, Rules, Examples, 58 OFF
The central nitrogen atom has sp 3 hybridization in the [nh 4] + ion. The electronic configuration of a nitrogen (n) atom is 1s 2 2s 2. According to vsepr theory we can find the hybridization of atoms in a molecule by the summation of the number of lone pairs of. The nitrogen atom is attached, to three h atoms.
MariePreAPChem Hybridization
According to vsepr theory we can find the hybridization of atoms in a molecule by the summation of the number of lone pairs of. The central nitrogen atom has sp 3 hybridization in the [nh 4] + ion. The nitrogen atom is attached, to three h atoms through three sigma covalent bonds and to one h^+ ion through one sigma..
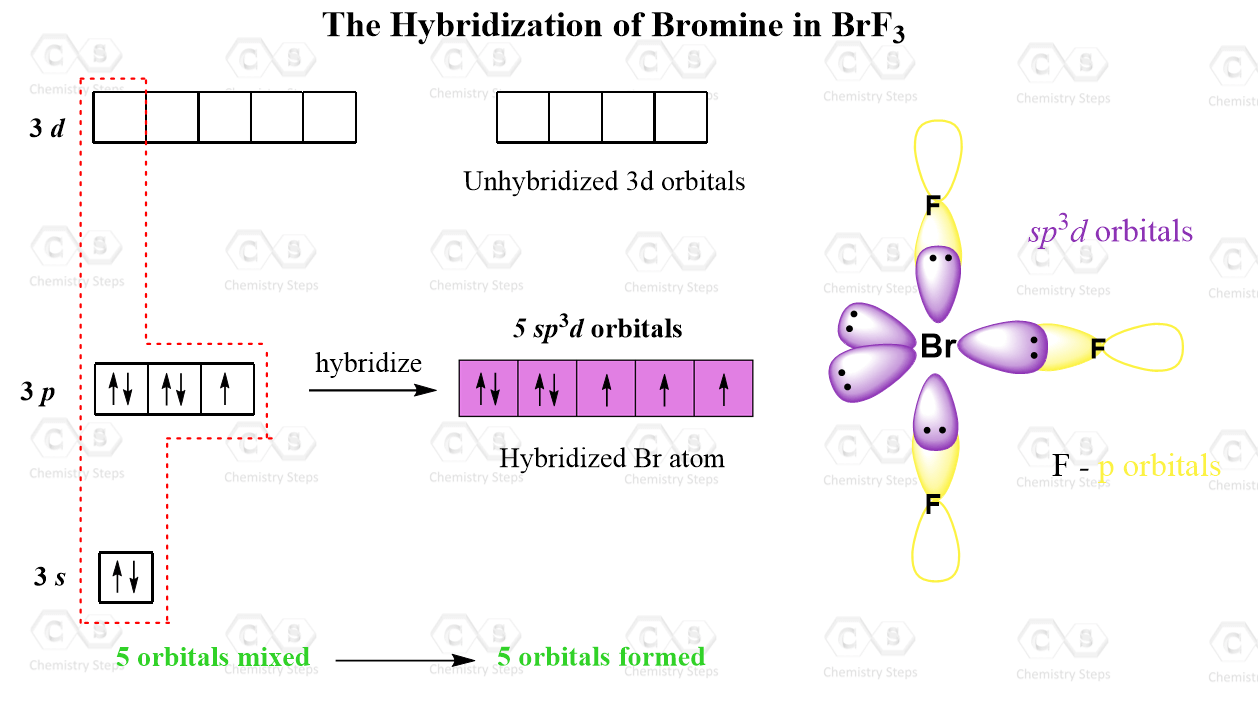
chemistry Hybridization In molecules containing double and triple bond.
The central nitrogen atom has sp 3 hybridization in the [nh 4] + ion. The nitrogen atom is attached, to three h atoms through three sigma covalent bonds and to one h^+ ion through one sigma. The electronic configuration of a nitrogen (n) atom is 1s 2 2s 2. According to vsepr theory we can find the hybridization of atoms.
Hybridization Chemistry
The nitrogen atom is attached, to three h atoms through three sigma covalent bonds and to one h^+ ion through one sigma. According to vsepr theory we can find the hybridization of atoms in a molecule by the summation of the number of lone pairs of. The central nitrogen atom has sp 3 hybridization in the [nh 4] + ion..
N atom in NH4+ involves the hybridization Filo
The central nitrogen atom has sp 3 hybridization in the [nh 4] + ion. According to vsepr theory we can find the hybridization of atoms in a molecule by the summation of the number of lone pairs of. The nitrogen atom is attached, to three h atoms through three sigma covalent bonds and to one h^+ ion through one sigma..
The Central Nitrogen Atom Has Sp 3 Hybridization In The [Nh 4] + Ion.
The nitrogen atom is attached, to three h atoms through three sigma covalent bonds and to one h^+ ion through one sigma. According to vsepr theory we can find the hybridization of atoms in a molecule by the summation of the number of lone pairs of. The electronic configuration of a nitrogen (n) atom is 1s 2 2s 2.

2]++4.+Tetrahedral.+sp3.+[Cd(NH3)4]2+.jpg)