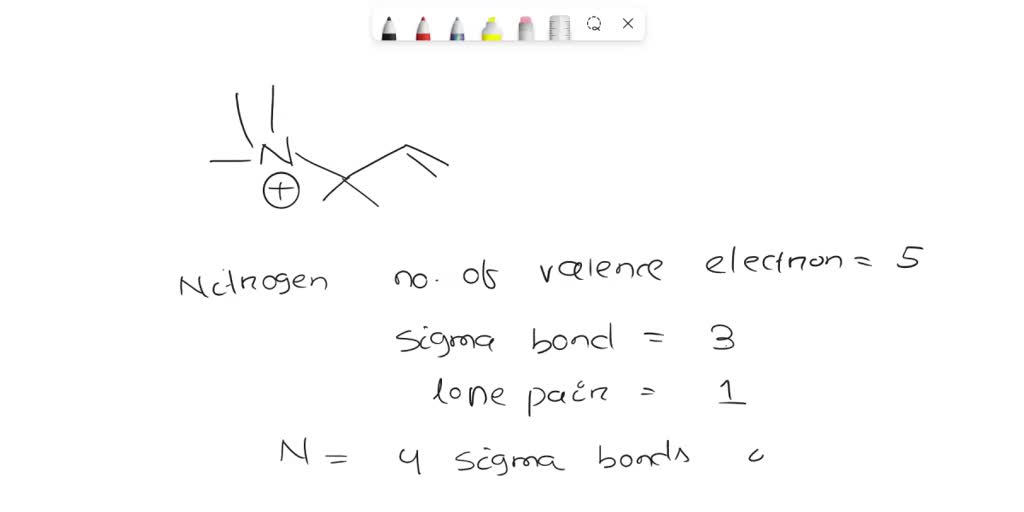
What Is The Hybridization Of The Nitrogen Atoms In N2
What Is The Hybridization Of The Nitrogen Atoms In N2 - What is the hybridization of the nitrogen atoms in each molecule? Nitrogen is frequently found in organic compounds. Nitrogen gas is shown below. In n2, both nitrogen atoms have a lone pair of electrons and three sigma bonds with the other nitrogen atom, forming an sp. It has a triple bond and one lone pair on each nitrogen atom.
Nitrogen gas is shown below. What is the hybridization of the nitrogen atoms in each molecule? In n2, both nitrogen atoms have a lone pair of electrons and three sigma bonds with the other nitrogen atom, forming an sp. Nitrogen is frequently found in organic compounds. It has a triple bond and one lone pair on each nitrogen atom.
What is the hybridization of the nitrogen atoms in each molecule? Nitrogen gas is shown below. It has a triple bond and one lone pair on each nitrogen atom. Nitrogen is frequently found in organic compounds. In n2, both nitrogen atoms have a lone pair of electrons and three sigma bonds with the other nitrogen atom, forming an sp.
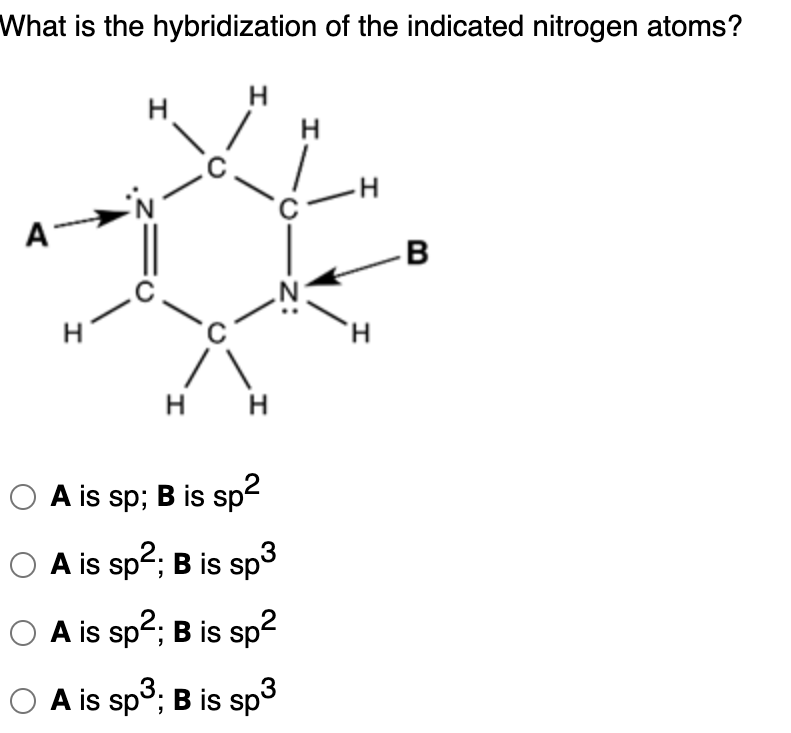
SOLVED Please identify the type of hybridization (sp, sp2, sp3) for
What is the hybridization of the nitrogen atoms in each molecule? In n2, both nitrogen atoms have a lone pair of electrons and three sigma bonds with the other nitrogen atom, forming an sp. It has a triple bond and one lone pair on each nitrogen atom. Nitrogen gas is shown below. Nitrogen is frequently found in organic compounds.
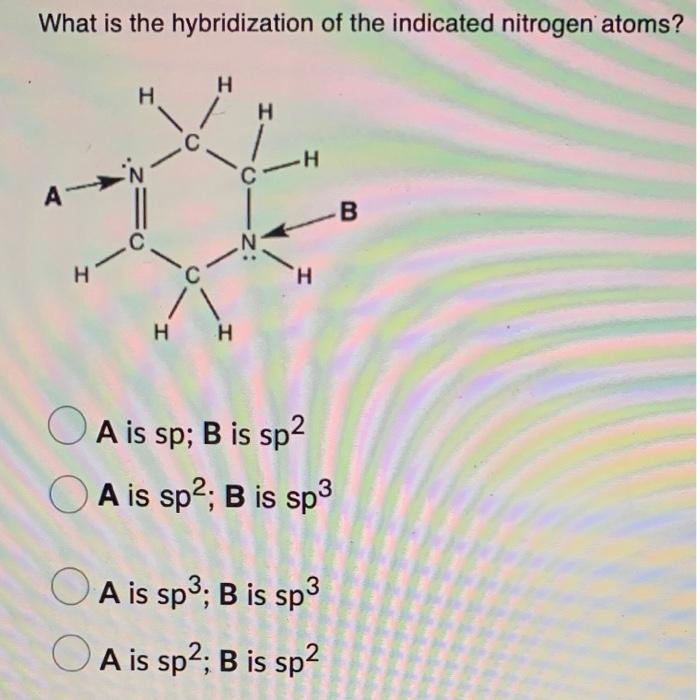
Solved What is the hybridization of the indicated nitrogen
In n2, both nitrogen atoms have a lone pair of electrons and three sigma bonds with the other nitrogen atom, forming an sp. It has a triple bond and one lone pair on each nitrogen atom. Nitrogen is frequently found in organic compounds. Nitrogen gas is shown below. What is the hybridization of the nitrogen atoms in each molecule?
Solved What is the hybridization of the indicated nitrogen
In n2, both nitrogen atoms have a lone pair of electrons and three sigma bonds with the other nitrogen atom, forming an sp. What is the hybridization of the nitrogen atoms in each molecule? Nitrogen is frequently found in organic compounds. It has a triple bond and one lone pair on each nitrogen atom. Nitrogen gas is shown below.
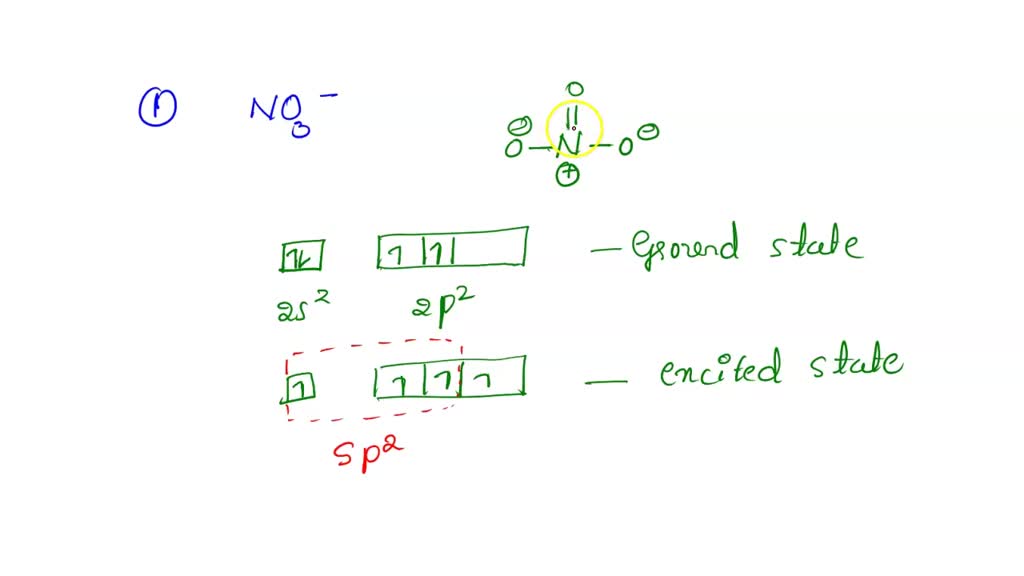
SOLVED What is the hybridization of nitrogen in the nitrite ion
What is the hybridization of the nitrogen atoms in each molecule? Nitrogen gas is shown below. Nitrogen is frequently found in organic compounds. It has a triple bond and one lone pair on each nitrogen atom. In n2, both nitrogen atoms have a lone pair of electrons and three sigma bonds with the other nitrogen atom, forming an sp.
[Solved] Identify how many N ( NITROGEN ) atoms have sp 3 hybridization
It has a triple bond and one lone pair on each nitrogen atom. In n2, both nitrogen atoms have a lone pair of electrons and three sigma bonds with the other nitrogen atom, forming an sp. What is the hybridization of the nitrogen atoms in each molecule? Nitrogen gas is shown below. Nitrogen is frequently found in organic compounds.
[Solved] Identify how many N (NITROGEN) atoms have sp hybridization for
Nitrogen is frequently found in organic compounds. Nitrogen gas is shown below. It has a triple bond and one lone pair on each nitrogen atom. In n2, both nitrogen atoms have a lone pair of electrons and three sigma bonds with the other nitrogen atom, forming an sp. What is the hybridization of the nitrogen atoms in each molecule?
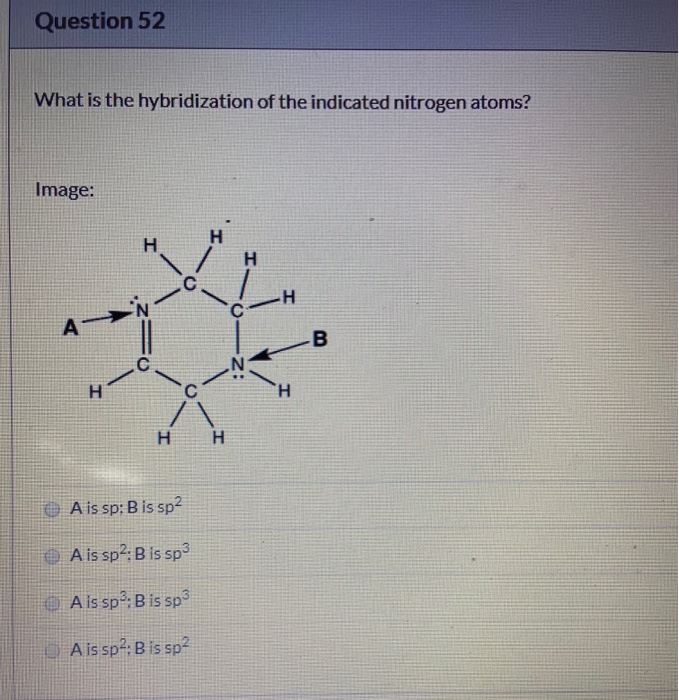
Solved Question 52 What Is The Hybridization Of The Indic...
Nitrogen is frequently found in organic compounds. Nitrogen gas is shown below. What is the hybridization of the nitrogen atoms in each molecule? In n2, both nitrogen atoms have a lone pair of electrons and three sigma bonds with the other nitrogen atom, forming an sp. It has a triple bond and one lone pair on each nitrogen atom.
[Solved] Identify how many N (NITROGEN) atoms have sp hybridization for
It has a triple bond and one lone pair on each nitrogen atom. Nitrogen is frequently found in organic compounds. In n2, both nitrogen atoms have a lone pair of electrons and three sigma bonds with the other nitrogen atom, forming an sp. Nitrogen gas is shown below. What is the hybridization of the nitrogen atoms in each molecule?
Solved Identify how many N (NITROGEN) atoms have \\(
It has a triple bond and one lone pair on each nitrogen atom. In n2, both nitrogen atoms have a lone pair of electrons and three sigma bonds with the other nitrogen atom, forming an sp. Nitrogen gas is shown below. What is the hybridization of the nitrogen atoms in each molecule? Nitrogen is frequently found in organic compounds.
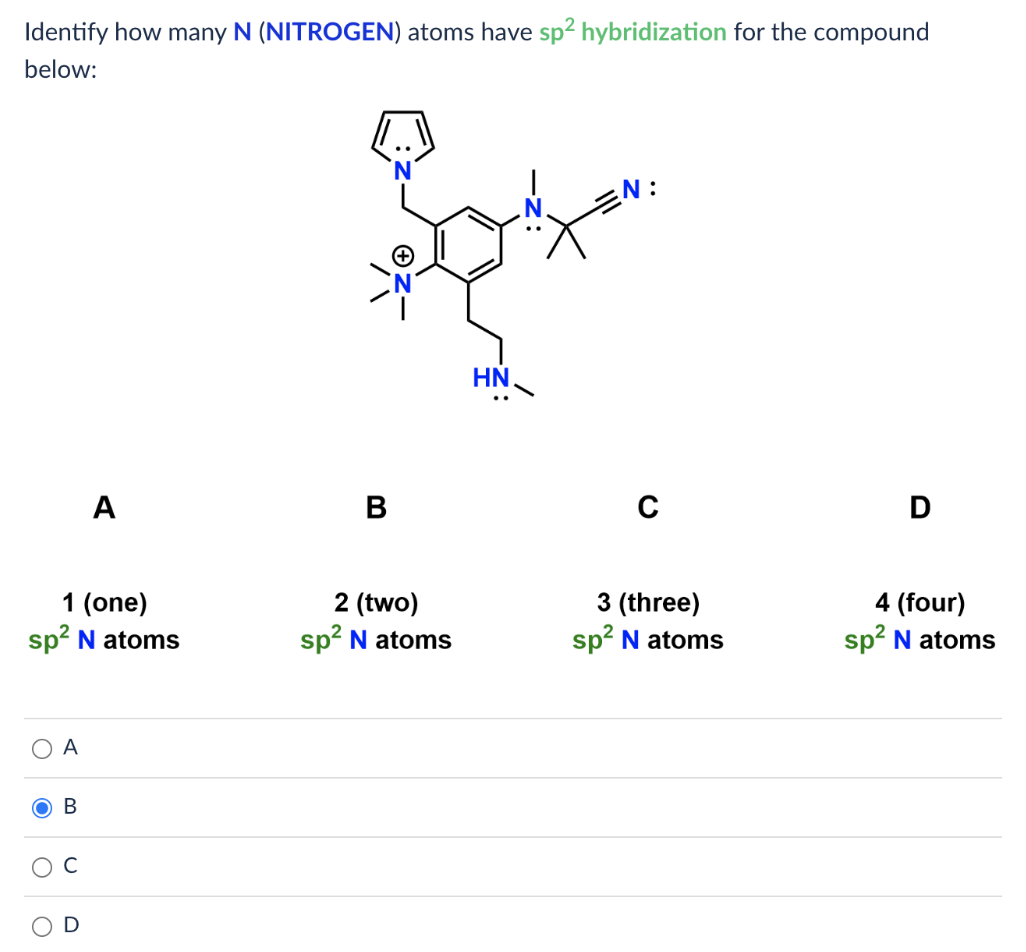
[Solved] Identify how many N (NITROGEN) atoms have sp2
What is the hybridization of the nitrogen atoms in each molecule? It has a triple bond and one lone pair on each nitrogen atom. Nitrogen is frequently found in organic compounds. In n2, both nitrogen atoms have a lone pair of electrons and three sigma bonds with the other nitrogen atom, forming an sp. Nitrogen gas is shown below.
Nitrogen Gas Is Shown Below.
Nitrogen is frequently found in organic compounds. In n2, both nitrogen atoms have a lone pair of electrons and three sigma bonds with the other nitrogen atom, forming an sp. What is the hybridization of the nitrogen atoms in each molecule? It has a triple bond and one lone pair on each nitrogen atom.
![[Solved] Identify how many N (NITROGEN) atoms have sp2](https://media.cheggcdn.com/media/250/25001471-53e2-40e1-97ef-668977575fc1/phppbkvdZ)