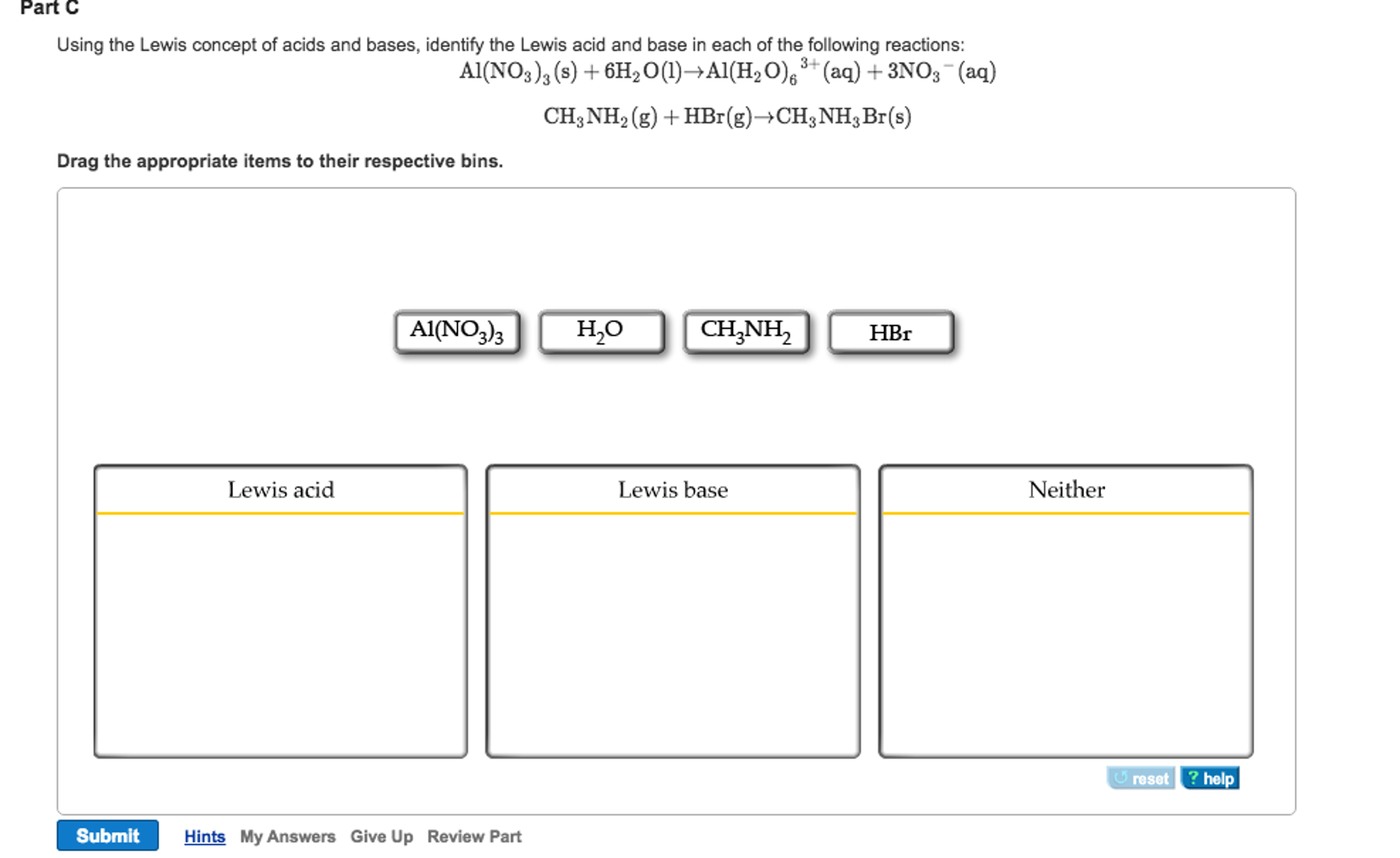
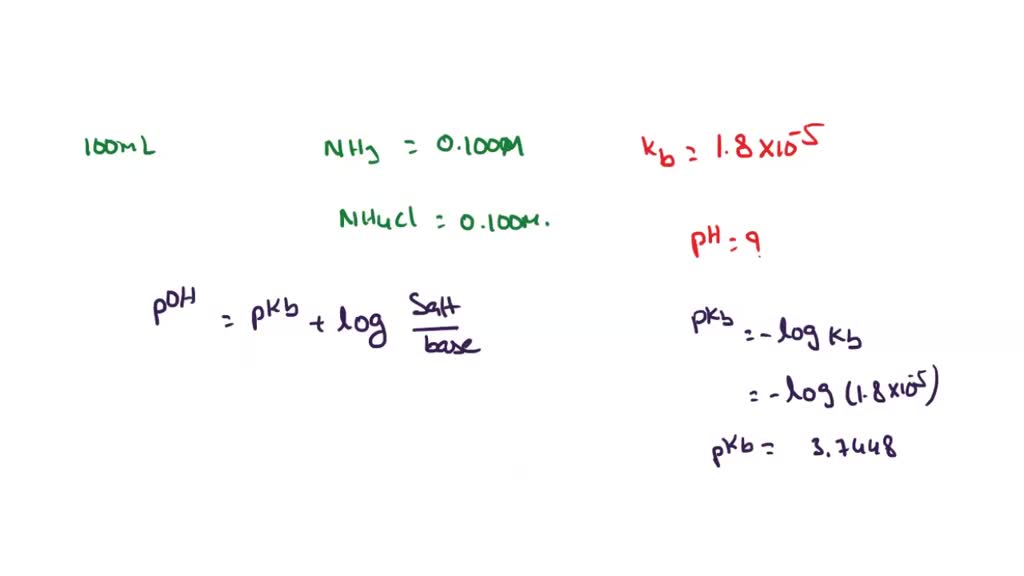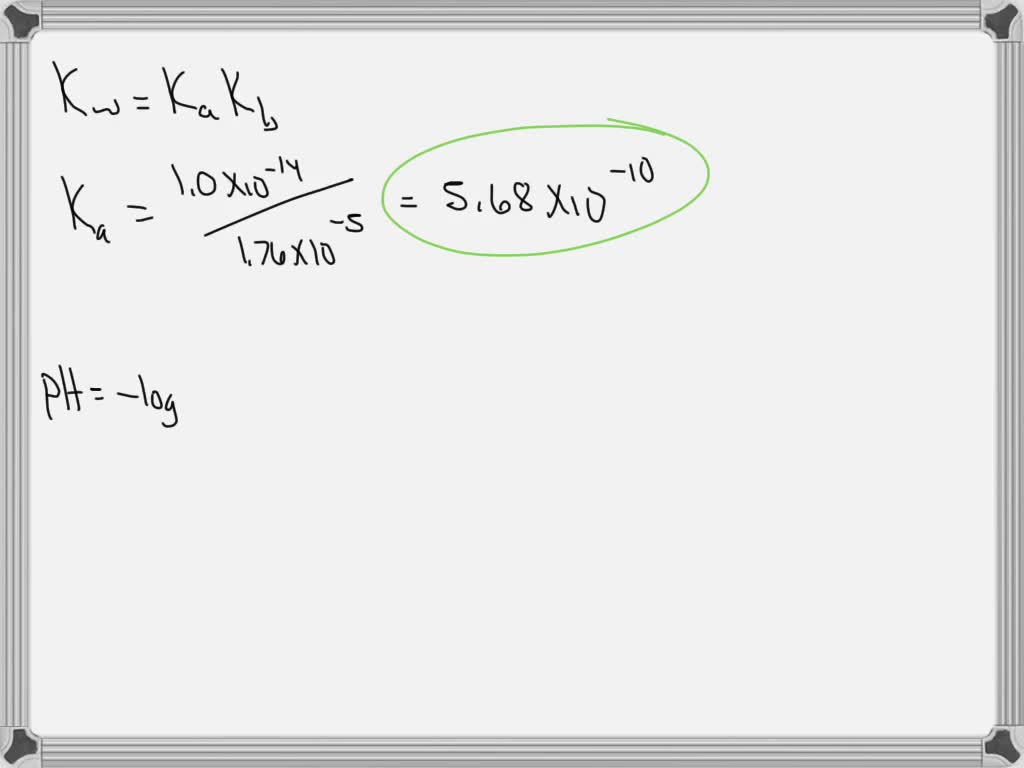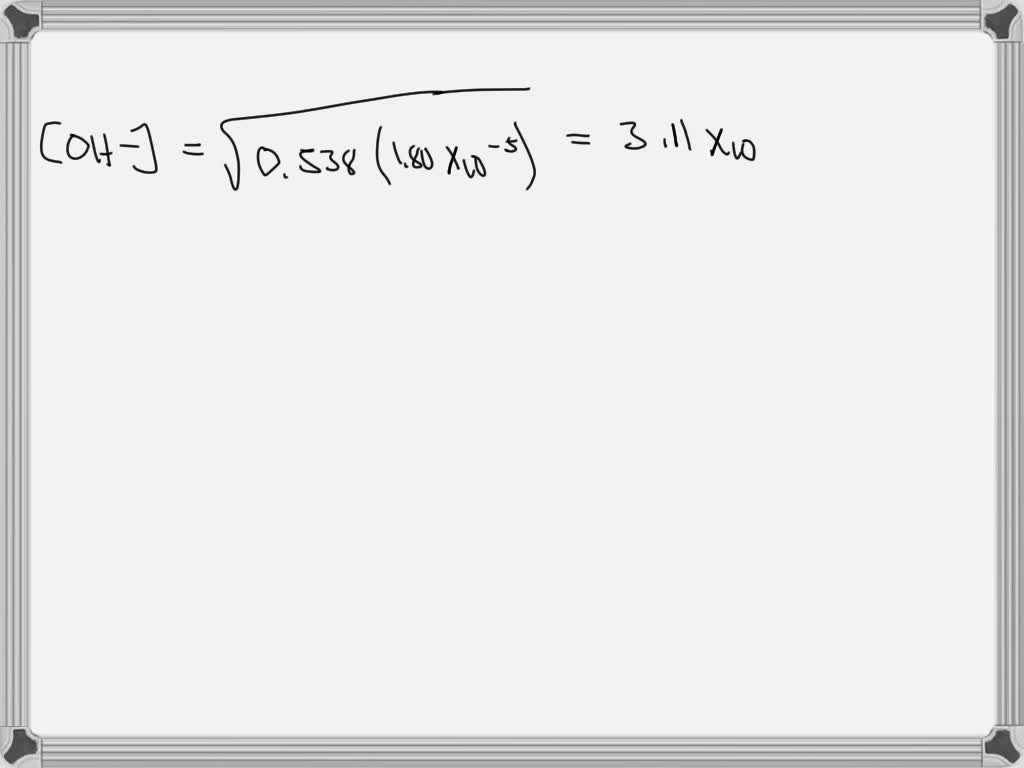What Is The Kb Of Nh3
What Is The Kb Of Nh3 - Acid with values less than one are considered weak. Read these instructions to learn how to use this acids and bases chart. Strong acids are listed at the top left hand corner of the table and have ka values >1 2. The table lists the k a values and the strength of each acid and base. Similarly, the equilibrium constant for the reaction of a weak base with. The dissociation reaction of nh3 in water is given. Acids with a k a value less than one are. The base dissociation constant, kb, for ammonia (nh3) provides a measure of its strength as a base in aqueous solution. For an aqueous solution of a weak acid, the dissociation constant is called the acid ionization constant (ka).
The base dissociation constant, kb, for ammonia (nh3) provides a measure of its strength as a base in aqueous solution. Acid with values less than one are considered weak. Acids with a k a value less than one are. The table lists the k a values and the strength of each acid and base. Similarly, the equilibrium constant for the reaction of a weak base with. Strong acids are listed at the top left hand corner of the table and have ka values >1 2. The dissociation reaction of nh3 in water is given. For an aqueous solution of a weak acid, the dissociation constant is called the acid ionization constant (ka). Read these instructions to learn how to use this acids and bases chart.
Acid with values less than one are considered weak. The base dissociation constant, kb, for ammonia (nh3) provides a measure of its strength as a base in aqueous solution. For an aqueous solution of a weak acid, the dissociation constant is called the acid ionization constant (ka). Read these instructions to learn how to use this acids and bases chart. The table lists the k a values and the strength of each acid and base. Similarly, the equilibrium constant for the reaction of a weak base with. The dissociation reaction of nh3 in water is given. Acids with a k a value less than one are. Strong acids are listed at the top left hand corner of the table and have ka values >1 2.
Solved C. Calculate The Kb For NH_3 Given That A 0.05 M S...
The base dissociation constant, kb, for ammonia (nh3) provides a measure of its strength as a base in aqueous solution. Similarly, the equilibrium constant for the reaction of a weak base with. Read these instructions to learn how to use this acids and bases chart. Acid with values less than one are considered weak. For an aqueous solution of a.
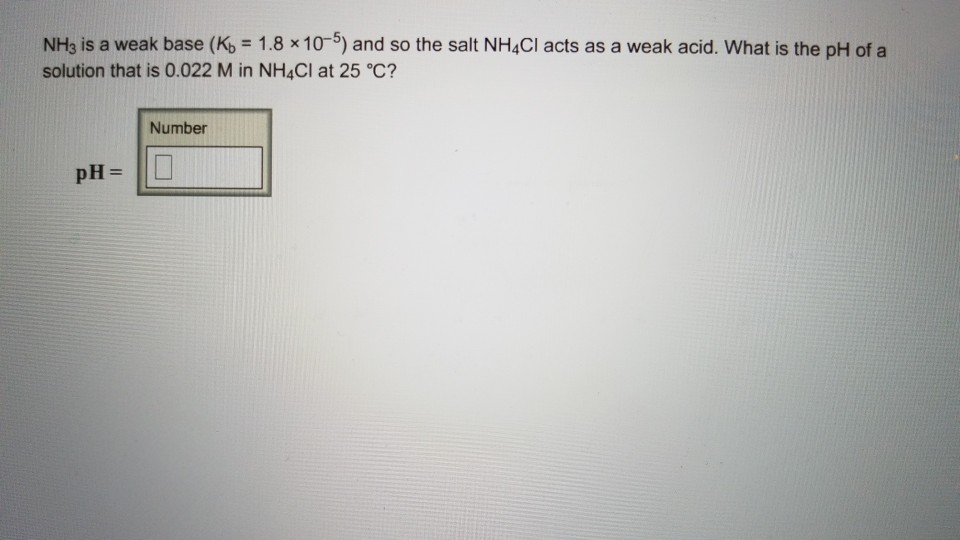
Solved NH3 is a weak base (Kb = 1.8 x 105) and so the salt
Acids with a k a value less than one are. The base dissociation constant, kb, for ammonia (nh3) provides a measure of its strength as a base in aqueous solution. Read these instructions to learn how to use this acids and bases chart. Acid with values less than one are considered weak. Similarly, the equilibrium constant for the reaction of.
Solved NH3 Kb = 1.8 x 105 CH3COOH HNO2 HCOOH НОСІ H2CO3 HF
The table lists the k a values and the strength of each acid and base. The dissociation reaction of nh3 in water is given. Acid with values less than one are considered weak. Acids with a k a value less than one are. For an aqueous solution of a weak acid, the dissociation constant is called the acid ionization constant.
SOLVED NH3 is a weak base with Kb = 1.8 x 105. A buffer is prepared
For an aqueous solution of a weak acid, the dissociation constant is called the acid ionization constant (ka). Strong acids are listed at the top left hand corner of the table and have ka values >1 2. Similarly, the equilibrium constant for the reaction of a weak base with. Acids with a k a value less than one are. The.
SOLVED Kb for ammonia (NH3) is 1.76 x 10^5. What is Ka for ammonium
Read these instructions to learn how to use this acids and bases chart. Acids with a k a value less than one are. The table lists the k a values and the strength of each acid and base. Strong acids are listed at the top left hand corner of the table and have ka values >1 2. For an aqueous.
SOLVED 'Determine the pH of a 0.420 M NH3 solution The Kb of NH3 is 1
For an aqueous solution of a weak acid, the dissociation constant is called the acid ionization constant (ka). The table lists the k a values and the strength of each acid and base. The dissociation reaction of nh3 in water is given. Similarly, the equilibrium constant for the reaction of a weak base with. Acid with values less than one.
[Solved] 10^2 M NH3 In Water Kb=1.9*10^5, What Is The Concentration
For an aqueous solution of a weak acid, the dissociation constant is called the acid ionization constant (ka). Read these instructions to learn how to use this acids and bases chart. Acids with a k a value less than one are. The table lists the k a values and the strength of each acid and base. Similarly, the equilibrium constant.
Solved 1) Ammonia, NH3, is a weak base with a Kb value of
Acid with values less than one are considered weak. The table lists the k a values and the strength of each acid and base. Acids with a k a value less than one are. The base dissociation constant, kb, for ammonia (nh3) provides a measure of its strength as a base in aqueous solution. Read these instructions to learn how.
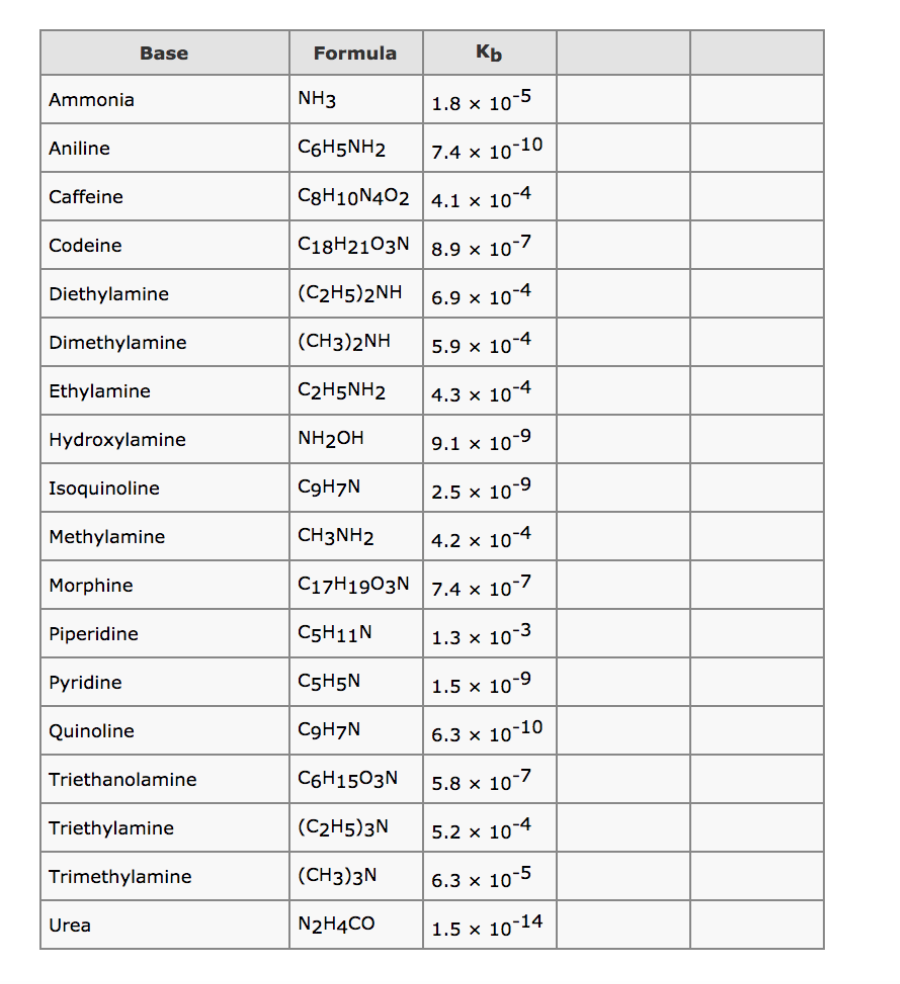
Solved Kb Formula NH3 ?6H5NH2 C8H10N4024.1 x 10 C18H2103N8.9
Acids with a k a value less than one are. For an aqueous solution of a weak acid, the dissociation constant is called the acid ionization constant (ka). The table lists the k a values and the strength of each acid and base. Read these instructions to learn how to use this acids and bases chart. Acid with values less.
Solved Ammonia, NH3, is a weak base with a Kb value of 1.8
For an aqueous solution of a weak acid, the dissociation constant is called the acid ionization constant (ka). The dissociation reaction of nh3 in water is given. Similarly, the equilibrium constant for the reaction of a weak base with. Strong acids are listed at the top left hand corner of the table and have ka values >1 2. Acid with.
The Base Dissociation Constant, Kb, For Ammonia (Nh3) Provides A Measure Of Its Strength As A Base In Aqueous Solution.
Strong acids are listed at the top left hand corner of the table and have ka values >1 2. For an aqueous solution of a weak acid, the dissociation constant is called the acid ionization constant (ka). The dissociation reaction of nh3 in water is given. Read these instructions to learn how to use this acids and bases chart.
Acids With A K A Value Less Than One Are.
The table lists the k a values and the strength of each acid and base. Similarly, the equilibrium constant for the reaction of a weak base with. Acid with values less than one are considered weak.