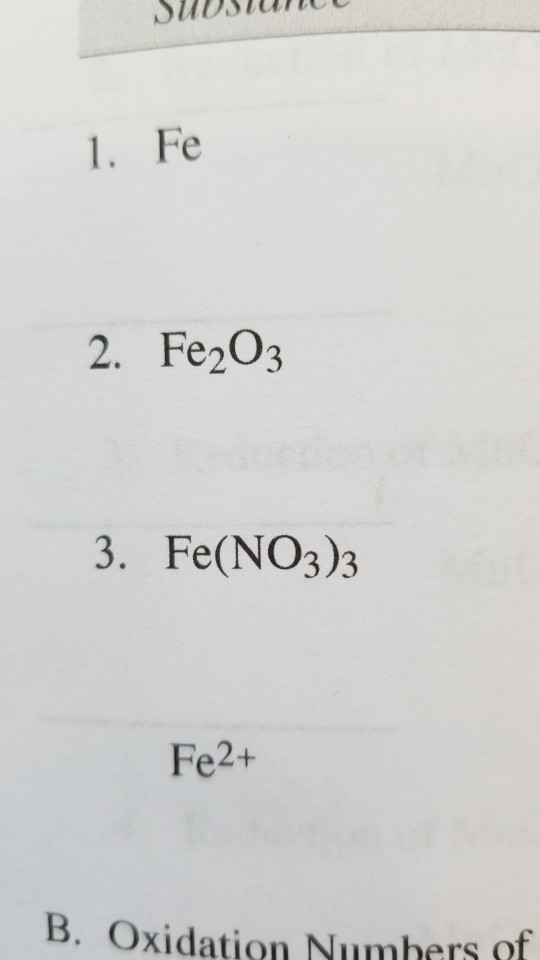What Is The Oxidation Number Of Iron In Fe2O3
What Is The Oxidation Number Of Iron In Fe2O3 - This means that each fe atom. The oxidation state of iron(iii) oxide is +3. The bond between the oxygen and iron atoms is an ionic bond, resulting from the difference in. The (ii) and (iii) are the oxidation states of the iron in the two compounds: The oxidation state of fe 2 o 3 is. That tells you that they contain fe 2. Fe 2 o 3 is the chemical formula of iron(iii) oxide which has three oxygen atoms, and two iron atoms.
The oxidation state of iron(iii) oxide is +3. The bond between the oxygen and iron atoms is an ionic bond, resulting from the difference in. The oxidation state of fe 2 o 3 is. This means that each fe atom. The (ii) and (iii) are the oxidation states of the iron in the two compounds: Fe 2 o 3 is the chemical formula of iron(iii) oxide which has three oxygen atoms, and two iron atoms. That tells you that they contain fe 2.
The oxidation state of fe 2 o 3 is. This means that each fe atom. The (ii) and (iii) are the oxidation states of the iron in the two compounds: That tells you that they contain fe 2. The oxidation state of iron(iii) oxide is +3. Fe 2 o 3 is the chemical formula of iron(iii) oxide which has three oxygen atoms, and two iron atoms. The bond between the oxygen and iron atoms is an ionic bond, resulting from the difference in.
how to calculate oxidation number of fe in feso4 (nh4)2so4 6h2o
The oxidation state of iron(iii) oxide is +3. This means that each fe atom. The oxidation state of fe 2 o 3 is. The (ii) and (iii) are the oxidation states of the iron in the two compounds: Fe 2 o 3 is the chemical formula of iron(iii) oxide which has three oxygen atoms, and two iron atoms.
Solved 1. Fe 2. Fe2O3 3. Fe(NO3)3 Fe2+ B. Oxidation Numbers
That tells you that they contain fe 2. The oxidation state of iron(iii) oxide is +3. The bond between the oxygen and iron atoms is an ionic bond, resulting from the difference in. This means that each fe atom. Fe 2 o 3 is the chemical formula of iron(iii) oxide which has three oxygen atoms, and two iron atoms.
Fe2o3 Oxidation Number
The (ii) and (iii) are the oxidation states of the iron in the two compounds: The oxidation state of iron(iii) oxide is +3. Fe 2 o 3 is the chemical formula of iron(iii) oxide which has three oxygen atoms, and two iron atoms. The oxidation state of fe 2 o 3 is. That tells you that they contain fe 2.
Calculate the OXIDATION NUMBER of Iron (Fe) in Fe3O4 YouTube
The oxidation state of fe 2 o 3 is. That tells you that they contain fe 2. Fe 2 o 3 is the chemical formula of iron(iii) oxide which has three oxygen atoms, and two iron atoms. The oxidation state of iron(iii) oxide is +3. The bond between the oxygen and iron atoms is an ionic bond, resulting from the.
Type of Reaction for Al + Fe2O3 = Al2O3 + Fe YouTube
Fe 2 o 3 is the chemical formula of iron(iii) oxide which has three oxygen atoms, and two iron atoms. The oxidation state of fe 2 o 3 is. The (ii) and (iii) are the oxidation states of the iron in the two compounds: The bond between the oxygen and iron atoms is an ionic bond, resulting from the difference.
Answered What is the oxidation number of iron,… bartleby
The (ii) and (iii) are the oxidation states of the iron in the two compounds: That tells you that they contain fe 2. The bond between the oxygen and iron atoms is an ionic bond, resulting from the difference in. The oxidation state of iron(iii) oxide is +3. Fe 2 o 3 is the chemical formula of iron(iii) oxide which.
For oxidation of iron,4Fe(s)+3O2 (g)→2Fe2 O3 (s)entropy change is 549
The oxidation state of iron(iii) oxide is +3. The (ii) and (iii) are the oxidation states of the iron in the two compounds: That tells you that they contain fe 2. This means that each fe atom. The bond between the oxygen and iron atoms is an ionic bond, resulting from the difference in.
What Is the Oxidation Number of Iron in Fe2o3 TalonwellFritz
The oxidation state of iron(iii) oxide is +3. The oxidation state of fe 2 o 3 is. That tells you that they contain fe 2. The bond between the oxygen and iron atoms is an ionic bond, resulting from the difference in. The (ii) and (iii) are the oxidation states of the iron in the two compounds:
Oxidation number of Fe (Iron) in Fe3O4// Redox reaction
The oxidation state of iron(iii) oxide is +3. The bond between the oxygen and iron atoms is an ionic bond, resulting from the difference in. The (ii) and (iii) are the oxidation states of the iron in the two compounds: That tells you that they contain fe 2. This means that each fe atom.
Fe 2 O 3 Is The Chemical Formula Of Iron(Iii) Oxide Which Has Three Oxygen Atoms, And Two Iron Atoms.
This means that each fe atom. The (ii) and (iii) are the oxidation states of the iron in the two compounds: The bond between the oxygen and iron atoms is an ionic bond, resulting from the difference in. The oxidation state of iron(iii) oxide is +3.
The Oxidation State Of Fe 2 O 3 Is.
That tells you that they contain fe 2.